


The article underscores seven essential GMP labeling practices that are pivotal for the success of clinical research. These include:
Such practices are not merely procedural; they are vital for ensuring compliance with regulatory standards, enhancing patient safety, and significantly mitigating the risk of costly errors. Consequently, these practices facilitate the swift and efficient market entry of medical innovations, underscoring their critical role in the Medtech landscape.
Navigating the complex landscape of Good Manufacturing Practice (GMP) labeling is essential for the success of clinical research in the pharmaceutical industry. As the demand for innovative medical solutions grows, understanding the intricacies of GMP compliance becomes vital for ensuring product safety and quality. With the stakes high—ranging from regulatory penalties to compromised patient trust—companies must effectively implement GMP labeling practices to safeguard their operations and enhance their market position. This article delves into the seven essential GMP labeling practices crucial for achieving clinical research success, offering insights that empower organizations to thrive in a competitive environment.
bioaccess® harnesses the regulatory efficiency of Latin America to bolster GMP adherence for Medtech innovations, significantly enhancing the clinical research environment. Ethical approvals are achieved in a remarkable 4-6 weeks, with enrollment accelerated by 50%. This empowers companies to adeptly navigate the complexities of GMP regulations. Such a swift method not only ensures that products meet the highest standards of quality and safety but also positions clients to capitalize on the expanding opportunities within the global medical technology market, valued at approximately $456 billion in 2021.
Industry leaders acknowledge the significance of regulatory agility; as one articulated, 'Speed in regulatory processes is crucial for bringing innovative solutions to market.'
Successful strategies for GMP labeling adherence encompass:
—elements that bioaccess® expertly facilitates. By merging its strengths with diverse patient pools in the Balkans and streamlined pathways in Australia, bioaccess® stands at the forefront of transforming clinical research, ensuring that Medtech innovations not only adhere to GMP labeling standards but also reach the market swiftly and efficiently.

GMP marking is essential in the pharmaceutical sector, serving as a cornerstone for item identification and traceability. Labels must effectively communicate critical information, including:
This practice not only ensures compliance with regulatory standards but also significantly enhances patient safety by providing clear and precise information about the products. Notably, approximately 8% of FDA recalls from 2012 to 2023 were associated with marking errors, underscoring the imperative for meticulous marking practices.
Adhering to GMP labeling is crucial, as it includes essential components such as:
All of which are integral to maintaining high safety standards. Moreover, effective GMP labeling practices can mitigate risks during clinical trials, ensuring that patients receive the correct medications and dosages, thereby fostering trust in pharmaceutical products. By implementing robust quality control measures and adhering to GMP guidelines, manufacturers can significantly diminish the likelihood of errors in product information, ultimately safeguarding patient health and bolstering the credibility of the pharmaceutical industry.

Regulatory requirements for GMP labeling are critical and vary significantly across different regions. In the United States, the FDA mandates that labels must prominently display essential information, including the name, strength, dosage, and expiration date. Adhering to these regulations is vital, not just to avoid legal repercussions but also to ensure consumer safety.
Non-compliance can result in severe consequences, such as product recalls and financial penalties, which can disrupt business operations. As regulatory scrutiny intensifies, it is imperative for businesses to actively monitor changes in GMP labeling regulations to ensure compliance and safeguard their market position.
Statistics reveal that organizations prioritizing GMP compliance experience markedly lower rates of regulatory violations, highlighting the necessity of a robust compliance strategy. Successful navigation of these regulations has been exemplified by companies that have implemented comprehensive training programs and established meticulous record-keeping systems, ensuring they effectively meet all marking requirements.

Non-compliance with GMP labeling standards can lead to severe consequences, including costly product recalls, legal penalties, and significant damage to a company's reputation. In 2023 alone, the FDA issued 599 Warning Letters, with 162 specifically related to current Good Manufacturing Practices (cGMP) violations. This underscores the critical need for adherence to these standards. For instance, Pfizer faced a substantial settlement exceeding $330 million due to inaccuracies associated with its hormone replacement therapy medication, Prempro, which resulted in adverse health effects and subsequent legal proceedings. Such incidents starkly illustrate the severe repercussions of inadequate labeling in the pharmaceutical industry, including potential contamination risks and variability in drug formulations stemming from non-compliance.
The impact of non-compliance extends beyond immediate financial losses; it can erode consumer trust and damage partnerships. Regulatory bodies, such as the FDA, actively oversee adherence, and violations can lead to warning letters, fines, or even criminal charges against responsible individuals. Legal experts emphasize that 'failure to meet GMP labeling standards can result in significant repercussions affecting a company’s operations, reputation, and financial performance,' particularly concerning recalls and legal penalties.
To mitigate these risks, companies must implement robust quality management systems that prioritize compliance and cultivate a culture of accountability. Continuous employee training and proactive risk management are essential to ensure adherence to GMP labeling standards, ultimately safeguarding public health and preserving the integrity of the pharmaceutical industry.

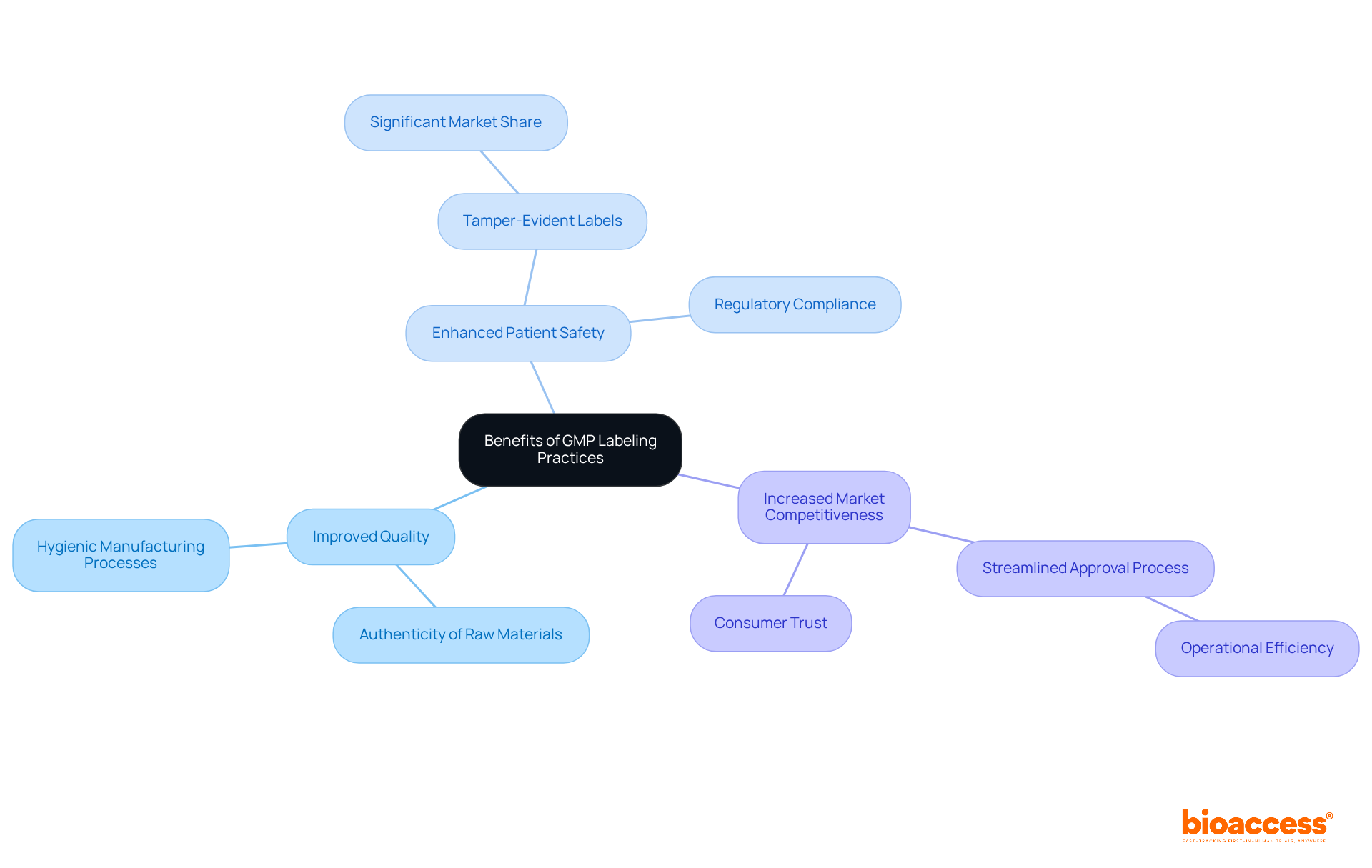
Adopting GMP marking practices yields substantial benefits, including improved quality of goods, enhanced patient safety, and increased market competitiveness. Precise and compliant identification fosters trust among consumers and regulatory bodies, a critical factor in the pharmaceutical industry.
For example, tamper-evident labels command a significant market share due to their effectiveness in preventing tampering, thereby bolstering patient safety and confidence in pharmaceutical products. Furthermore, efficient tagging practices streamline the approval process, aligning with the projected expansion of the global pharmaceutical tagging market, anticipated to grow at a CAGR of approximately 5% from 2023 to 2028. This efficiency not only facilitates smoother product launches but also positions companies advantageously within a competitive landscape.
Industry leaders, such as Arne Reis, emphasize that adherence to GMP labeling standards is essential for maintaining high-quality production and ensuring compliance with international regulations, ultimately fostering a more reliable and trusted market presence. Additionally, educating staff involved in the production process is vital for ensuring compliance with GMP standards.
By prioritizing these practices, companies can significantly enhance their operational efficiency and market positioning, ensuring their products consistently meet the highest standards of safety and efficacy.
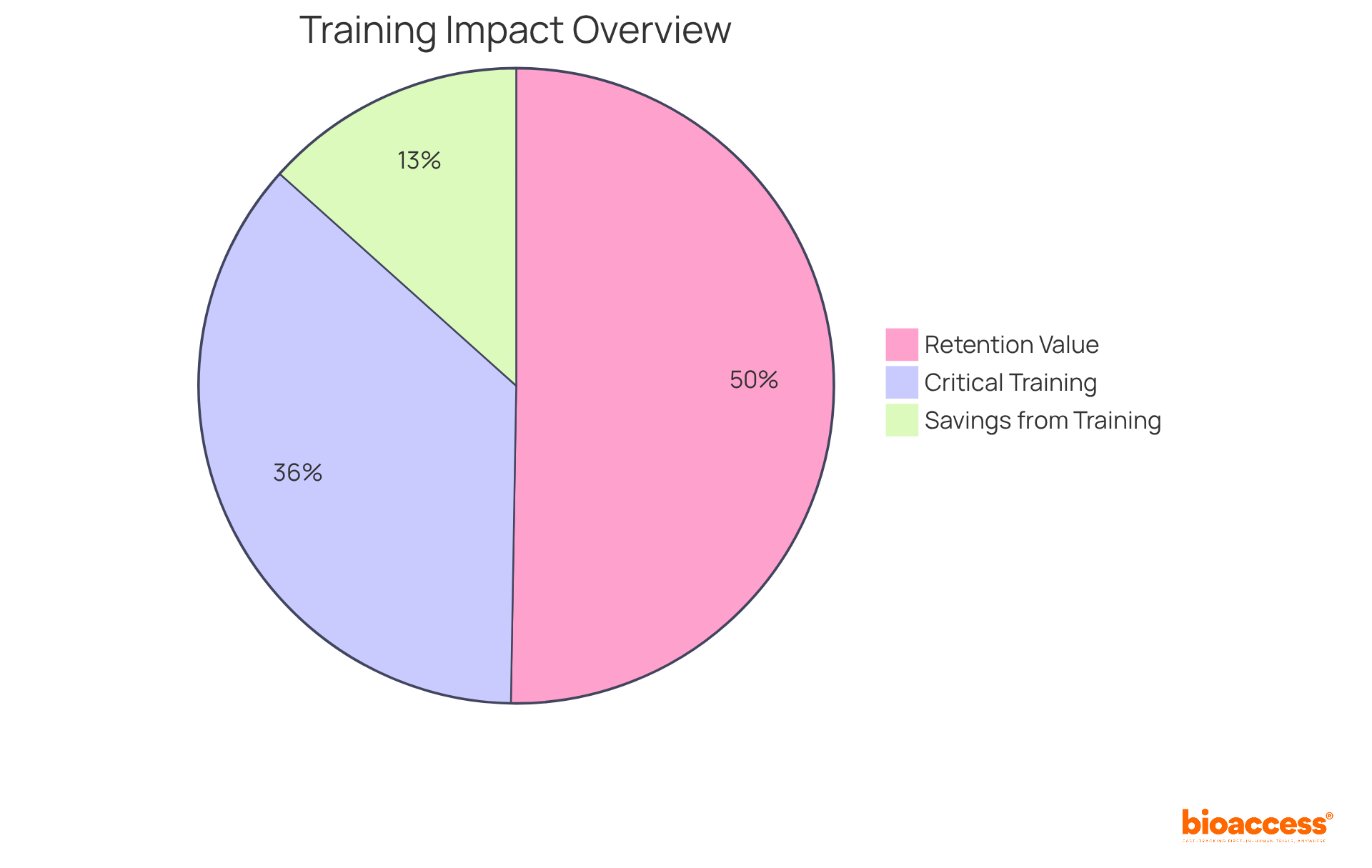
Training and education are paramount for achieving GMP certification. Organizations must prioritize comprehensive training programs that provide employees with a robust understanding of regulatory requirements and best practices for GMP labeling. Regular training sessions not only reinforce GMP principles but also significantly minimize errors and cultivate a culture of adherence within the organization. This proactive approach not only enhances product quality but also equips staff for successful audits and inspections.
Statistics indicate that companies investing in structured training programs see a notable increase in adherence rates, with 68% of employees asserting that training and development is the most critical policy of their organization. Furthermore, organizations that implement stringent training programs can save an average of $2.54 million, underscoring the financial benefits of investing in employee education. Additionally, 94% of employees express that they would remain with a company longer if it prioritized their learning and development.
Companies are also twice as likely to report improved employee retention rates when utilizing an education platform designed for diverse audiences (62%) compared to one aimed solely at employees (30%). By nurturing a well-trained workforce, companies can ensure compliance with GMP labeling standards and sustain high-quality product outputs.
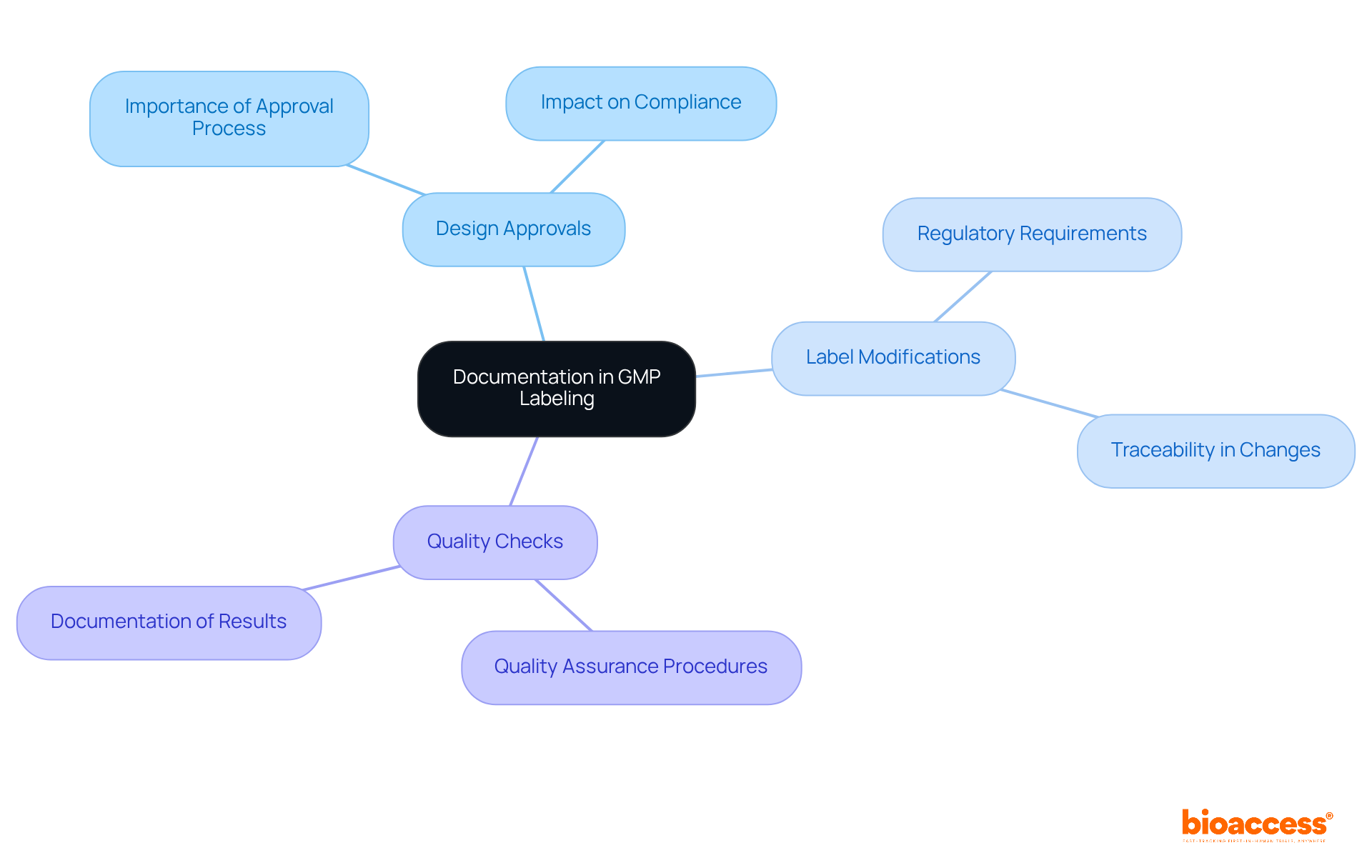
Documentation serves as a cornerstone of GMP tagging, acting as a vital record of adherence and quality assurance. Companies must uphold meticulous records of marking processes, which encompass design approvals, label modifications, and quality checks. This documentation not only bolsters regulatory compliance but also establishes a foundation for traceability during product recalls or audits. A robust documentation system is essential for demonstrating compliance with GMP standards and protecting the integrity of GMP labeling practices.
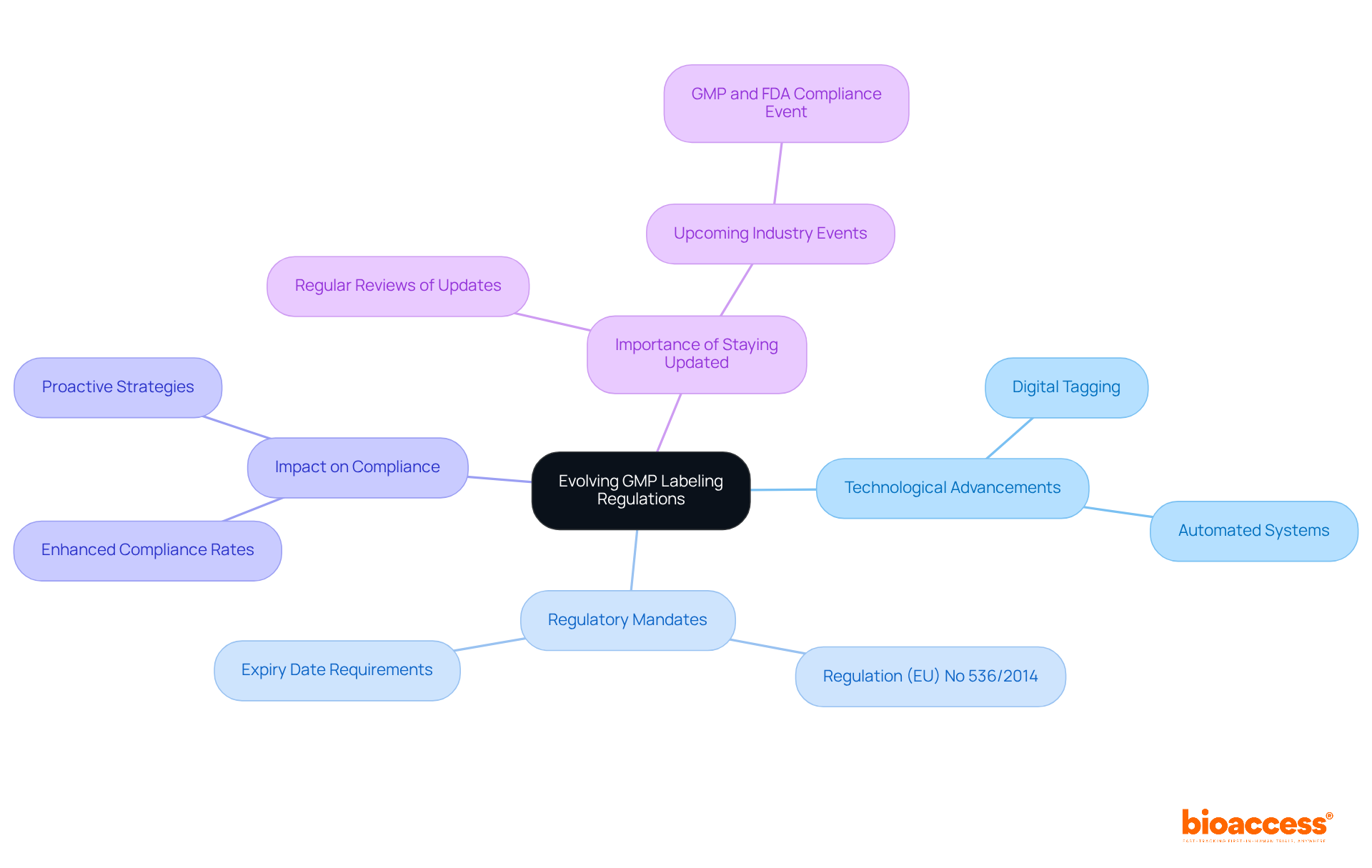
GMP marking regulations are in a state of constant evolution, influenced by technological advancements and shifts in regulatory frameworks. Staying abreast of these changes is crucial for companies aiming to maintain compliance. Experts like Ana Criado, Director of Regulatory Affairs and CEO of Mahu Pharma, emphasize the importance of understanding these regulations, particularly in the context of Colombia's unique regulatory landscape. The rise of digital tagging and automated systems is revolutionizing label management and application processes, leading to improved accuracy and efficiency.
For instance, Regulation (EU) No 536/2014 mandates specific marking requirements for Investigational Medicinal Products (IMPs), including the necessity of including the name of the substance on packaging in blind clinical trials. Additionally, the expiry date or re-test date must be provided on the primary packaging in month and year format. Organizations are encouraged to conduct regular reviews of regulatory updates and adjust their marking practices accordingly. This proactive strategy not only guarantees adherence but also boosts competitiveness in the market.
Companies that have incorporated digital solutions into their tagging processes have reported significant enhancements in compliance rates, demonstrating the tangible benefits of embracing technology in GMP labeling. As noted by Bermingham, Castleman & Pierce Inc., 'Quality is never an accident; it is always the result of high intention, sincere effort, intelligent direction, and skillful execution.' Furthermore, with the GMP and FDA Compliance in Pharmaceutical Development and IMP Manufacturing event scheduled for April 2026, it is essential for professionals to stay informed about industry developments, a sentiment echoed by regulatory experts like Katherine Ruiz, who specializes in regulatory affairs for medical devices and in vitro diagnostics in Colombia.
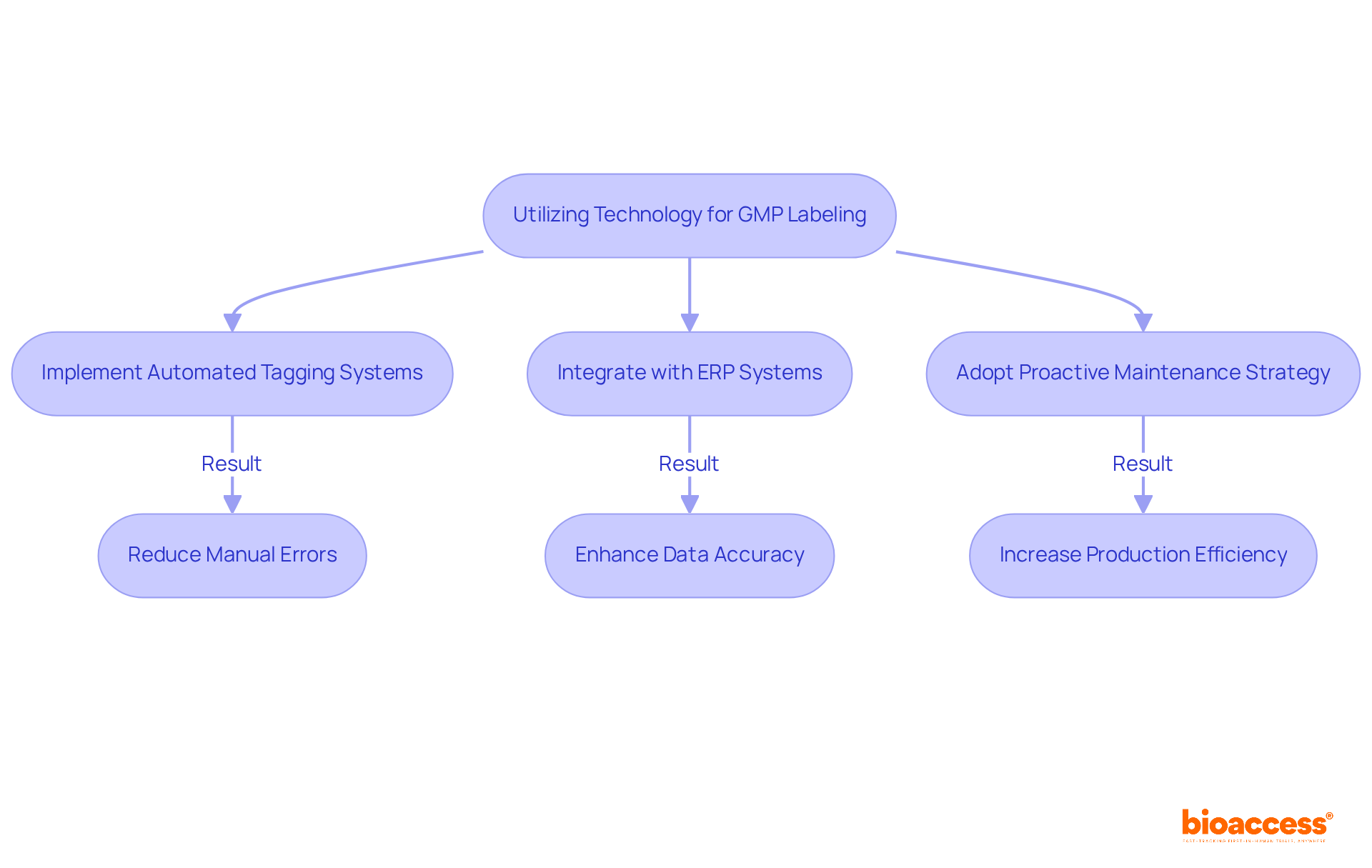
Utilizing technology is essential for streamlining GMP labeling processes. Automated tagging systems significantly minimize manual errors and guarantee that GMP labeling is consistently applied in accordance with regulatory requirements. Companies employing automatic machines report an impressive 85% reduction in marking errors compared to traditional manual methods. Moreover, integrating tagging software with enterprise resource planning (ERP) systems enhances data accuracy and facilitates real-time updates to tagging information. By embracing these technologies, organizations can boost efficiency, mitigate compliance risks, and elevate the overall quality of their offerings, particularly through GMP labeling.
Additionally, implementing a proactive maintenance strategy is vital for maximizing the lifespan and performance of automatic labelers. Recent advancements in automated tagging technology have shown that organizations can enhance production efficiency by 25% to 40%, while also improving overall quality. For instance, a cosmetic production facility doubled its yearly output by adopting an automatic tagging machine, illustrating the tangible benefits of these innovations. By leveraging these advancements, companies can enhance operational efficiency, reduce regulatory risks, and ultimately deliver superior products to the market.
Organizations must adopt several best practices to achieve effective GMP labeling compliance.
Regularly Review and Update Procedures: It is crucial to ensure that marking procedures consistently align with current regulations, thereby avoiding compliance issues. As W. Edwards Deming articulated, "Quality comes not from inspection, but from improvement of the production process," underscoring the necessity for continuous updates.
Implement a Robust Quality Management System (QMS): Establishing a QMS that incorporates checks and balances is essential for enhancing accuracy in product identification and traceability. The improvement of quality inherently boosts productivity, thereby making a compelling case for a well-structured QMS.
Foster a Culture of Compliance: Organizations should encourage continuous training and education for all personnel engaged in tagging processes. This commitment to learning strengthens the significance of adherence and empowers employees to uphold quality standards. Companies like Toyota exemplify successful implementation of such cultures, resulting in significant enhancements in quality and efficiency.
Utilize Technology: Leveraging automation and digital tools can streamline tagging operations effectively. Technology significantly enhances consistency and accuracy, thereby reducing the risk of human error. Data-driven decision-making is vital for optimizing these processes, ensuring that organizations can adapt and improve continuously.
By adhering to these best practices, companies can fortify their compliance efforts, ensuring the safety and quality of their products while adeptly navigating the complexities associated with GMP labeling.

The significance of GMP labeling practices in clinical research is paramount. These practices not only ensure compliance with regulatory standards but also enhance the overall quality and safety of pharmaceutical products. By adopting robust strategies for GMP labeling, organizations can adeptly navigate the complexities of regulatory requirements while fostering trust among consumers and stakeholders.
Throughout this discussion, essential aspects such as thorough documentation, regular audits, proactive engagement with regulatory bodies, and the integration of technology have been highlighted as critical components for successful GMP labeling. The consequences of non-compliance, including costly recalls and legal ramifications, further underscore the necessity of adhering to these standards. Moreover, the benefits of implementing effective GMP labeling practices extend beyond mere compliance; they contribute to improved product quality, enhanced patient safety, and increased competitiveness in the market.
In conclusion, prioritizing GMP labeling transcends regulatory obligation; it is a strategic imperative for organizations within the pharmaceutical industry. By investing in training, adopting technology, and fostering a culture of compliance, companies can ensure their products meet the highest standards of safety and efficacy. Embracing these practices will ultimately safeguard public health and strengthen the integrity of the pharmaceutical sector, paving the way for continued innovation and success in an evolving regulatory landscape.
What is bioaccess® and how does it benefit Medtech innovations?
bioaccess® leverages the regulatory efficiency of Latin America to enhance GMP compliance for Medtech innovations, achieving ethical approvals in 4-6 weeks and accelerating enrollment by 50%.
Why is regulatory agility important in the Medtech industry?
Regulatory agility is crucial for bringing innovative solutions to market quickly, which is essential for companies to capitalize on the expanding global medical technology market, valued at approximately $456 billion in 2021.
What strategies does bioaccess® use to ensure GMP labeling adherence?
Successful strategies for GMP labeling adherence include thorough documentation, regular audits, and proactive engagement with regulatory bodies, all of which bioaccess® expertly facilitates.
How does GMP labeling contribute to patient safety in pharmaceuticals?
GMP labeling ensures compliance with regulatory standards and enhances patient safety by providing clear information about active ingredients, usage instructions, and storage conditions.
What are the consequences of marking errors in pharmaceuticals?
Approximately 8% of FDA recalls from 2012 to 2023 were associated with marking errors, highlighting the importance of meticulous marking practices to prevent such issues.
What are the key components of GMP labeling?
Key components of GMP labeling include quality assurance, monitoring, and documentation, which are essential for maintaining high safety standards.
What are the regulatory requirements for GMP labeling in the United States?
In the U.S., the FDA mandates that labels display essential information such as the name, strength, dosage, and expiration date prominently.
What can happen if a company fails to comply with GMP labeling regulations?
Non-compliance can lead to severe consequences, including product recalls and financial penalties, which can disrupt business operations.
How can organizations improve their GMP compliance?
Organizations can improve their GMP compliance by implementing comprehensive training programs and establishing meticulous record-keeping systems to meet all marking requirements effectively.