


The landscape of biologics in Bulgaria is evolving rapidly, creating an urgent need for a robust framework for post-market safety monitoring. This essential process not only guarantees the ongoing effectiveness and safety of biological products but also plays a crucial role in protecting public health and fostering trust in these therapies. However, the integration of diverse data sources and the adaptation to changing regulatory requirements pose significant challenges. How can organizations effectively navigate these hurdles to enhance safety monitoring practices and ultimately improve patient outcomes?
Addressing these challenges requires a strategic approach that leverages data and collaboration. By establishing a comprehensive safety monitoring system, stakeholders can ensure that biological products remain safe and effective throughout their lifecycle. This proactive stance not only mitigates risks but also reinforces the credibility of the biologics sector. As we delve deeper into the Medtech landscape, it becomes clear that bioaccess plays a pivotal role in overcoming these key challenges, paving the way for improved patient outcomes.
The process of post-market safety monitoring for biologics in Bulgaria is critical and involves systematically collecting, analyzing, and interpreting data on the effectiveness and risks of biological products after they receive market approval. This oversight is essential for identifying any adverse effects or unexpected outcomes that may emerge once the product is widely used. It includes various activities such as:
Efficient post-market health monitoring ensures that concerns are swiftly addressed, safeguarding patient well-being and maintaining public trust in biologics.
In a global context, regulatory agencies like INVIMA in Colombia play a vital role in monitoring the quality of health products. Classified as a Level 4 health authority by the Pan American Health Organization/World Health Organization, INVIMA is responsible for inspecting and supervising the marketing and manufacturing of health products, including medical devices. This classification underscores the importance of robust regulatory frameworks in ensuring the safety and efficacy of biologics, a crucial consideration for clinical research professionals.
As we navigate the complexities of the Medtech landscape, it is imperative to recognize the role of bioaccess in addressing key challenges. Collaboration among stakeholders is essential to enhance post-market safety monitoring for biologics in Bulgaria and to ensure that these products continue to meet safety and effectiveness standards. The next steps involve fostering partnerships and sharing insights to strengthen regulatory practices and improve patient outcomes.
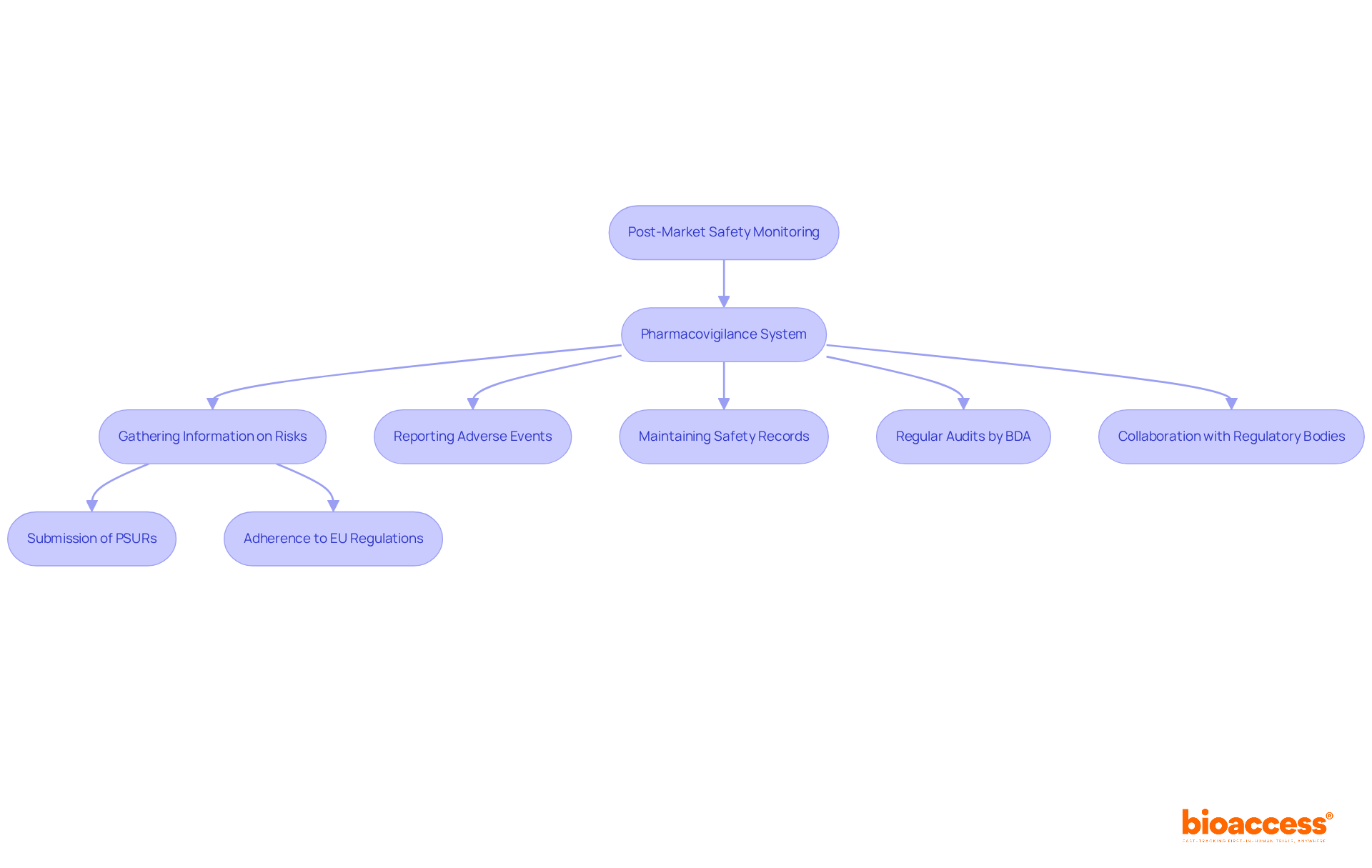
In Bulgaria, post-market safety monitoring for biologics is crucial for ensuring patient safety and product efficacy. Regulated by the Bulgarian Drug Agency (BDA) and aligned with European Medicines Agency (EMA) guidelines, this process is essential for maintaining high standards in clinical research.
A robust pharmacovigilance system is a fundamental requirement, actively gathering and assessing information regarding potential risks associated with biologics. This includes the submission of Periodic Safety Update Reports (PSURs) and strict adherence to the EU's pharmacovigilance regulations. Businesses must ensure they have effective systems in place for reporting adverse events and maintaining comprehensive records of all safety-related information.
Regular audits and inspections by the BDA further reinforce compliance with these regulatory requirements, ensuring that the highest standards are upheld. As the Medtech landscape evolves, collaboration and proactive engagement with regulatory bodies become increasingly vital for navigating challenges in clinical research.
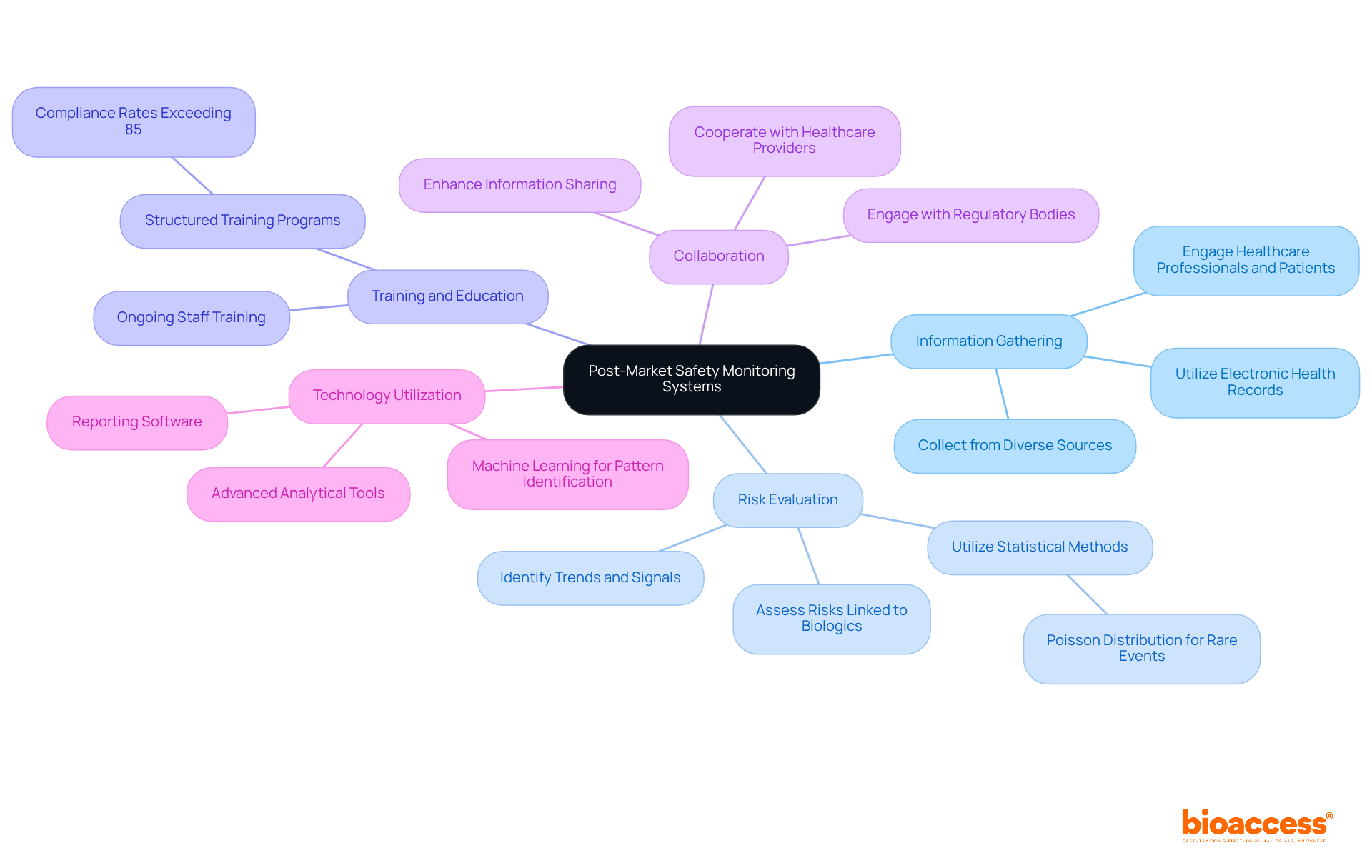
To implement effective post-market oversight monitoring systems, organizations must adopt a multi-faceted approach that includes:
As Geoffrey Moore highlighted, 'information is essential for making intelligent business choices,' stressing the significance of information in after-sales oversight.
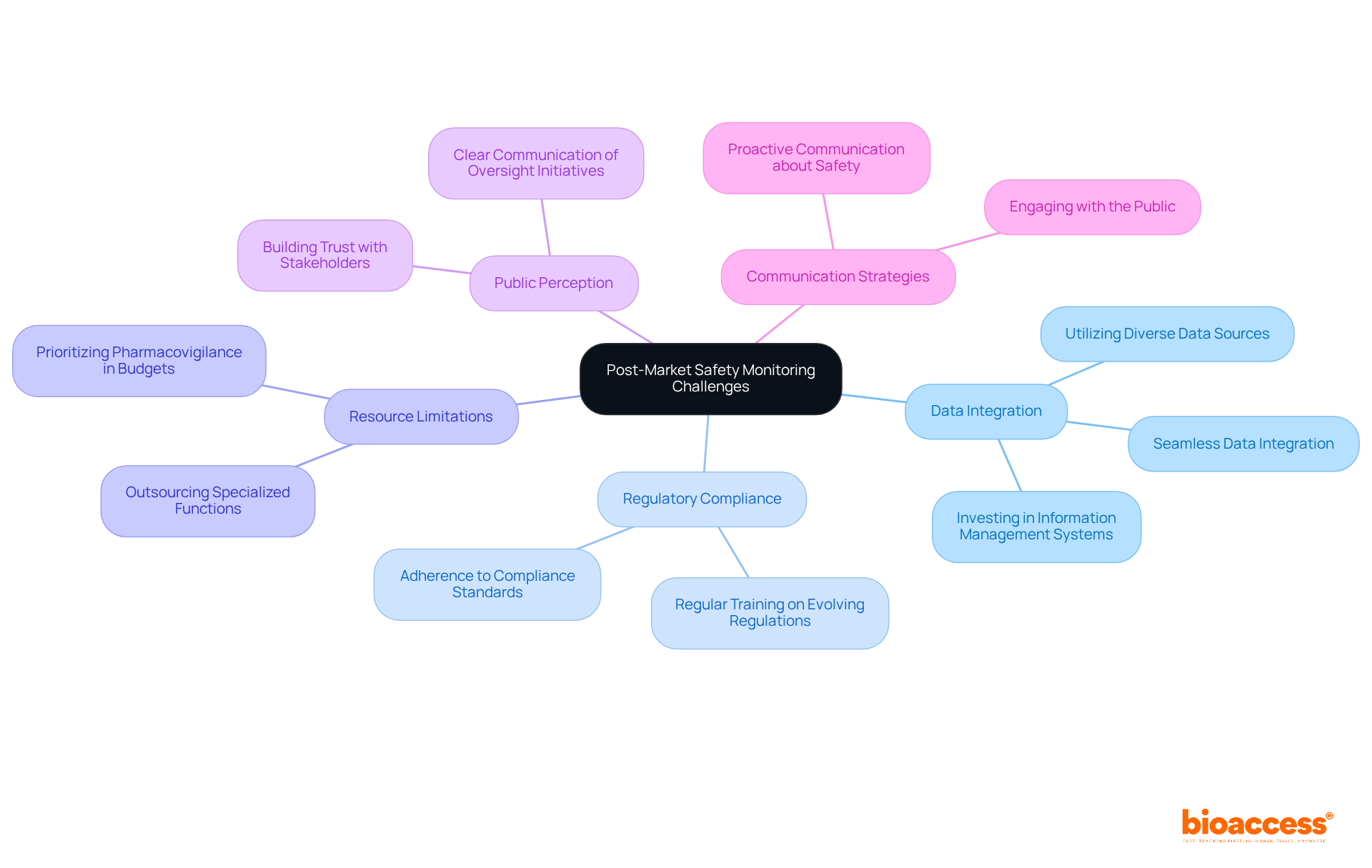
Organizations face significant challenges in post-market safety monitoring for biologics in Bulgaria, especially when it comes to integrating information from diverse sources. This integration is crucial for conducting thorough risk evaluations. Investing in robust information management systems is essential, as these systems enable seamless data integration and improve the accuracy of safety assessments. For instance, the FDA collects around 1.5 million adverse drug event (ADE) reports each year, highlighting the urgent need for effective data integration in pharmacovigilance.
Regulatory compliance adds another layer of complexity, as organizations must keep pace with evolving regulations. Regular training and updates on these changes are vital to ensure adherence. Additionally, limited resources can restrict oversight capabilities. Therefore, prioritizing pharmacovigilance in budget allocations and considering outsourcing certain functions to specialized firms can be beneficial.
Public perception of safety can complicate matters further, as negative opinions may impede product adoption. Clear communication about oversight initiatives and their outcomes is essential for building trust with stakeholders. As C Lee Ventola emphasizes, integrating evidence from all available sources is critical for safeguarding public health interests. Moreover, with the biologics sector projected to grow at a compound annual growth rate of 15% until 2027, the importance of post-market safety monitoring for biologics in Bulgaria is increasingly paramount. By tackling these challenges through strategic data integration and proactive communication, organizations can significantly enhance post-market safety monitoring for biologics in Bulgaria.
The significance of post-market safety monitoring for biologics in Bulgaria is paramount. This essential process not only ensures the ongoing evaluation of biological products but also facilitates the identification and management of any adverse effects that may arise post-market approval. By implementing robust monitoring systems, organizations can effectively protect patient safety and uphold public confidence in biologics.
Throughout this discussion, several key aspects of effective post-market safety monitoring have been underscored. These include:
Addressing challenges such as data integration, regulatory compliance, and public perception is crucial for enhancing the safety monitoring framework.
As the biologics sector continues to expand, the necessity for diligent post-market safety monitoring practices becomes increasingly critical. Organizations must prioritize these strategies, invest in effective systems, and engage in open communication with both the public and regulatory bodies. By doing so, they will not only ensure compliance with regulatory standards but also contribute to the overall safety and efficacy of biologics, ultimately benefiting patients and healthcare systems alike.
What is post-market safety monitoring for biologics?
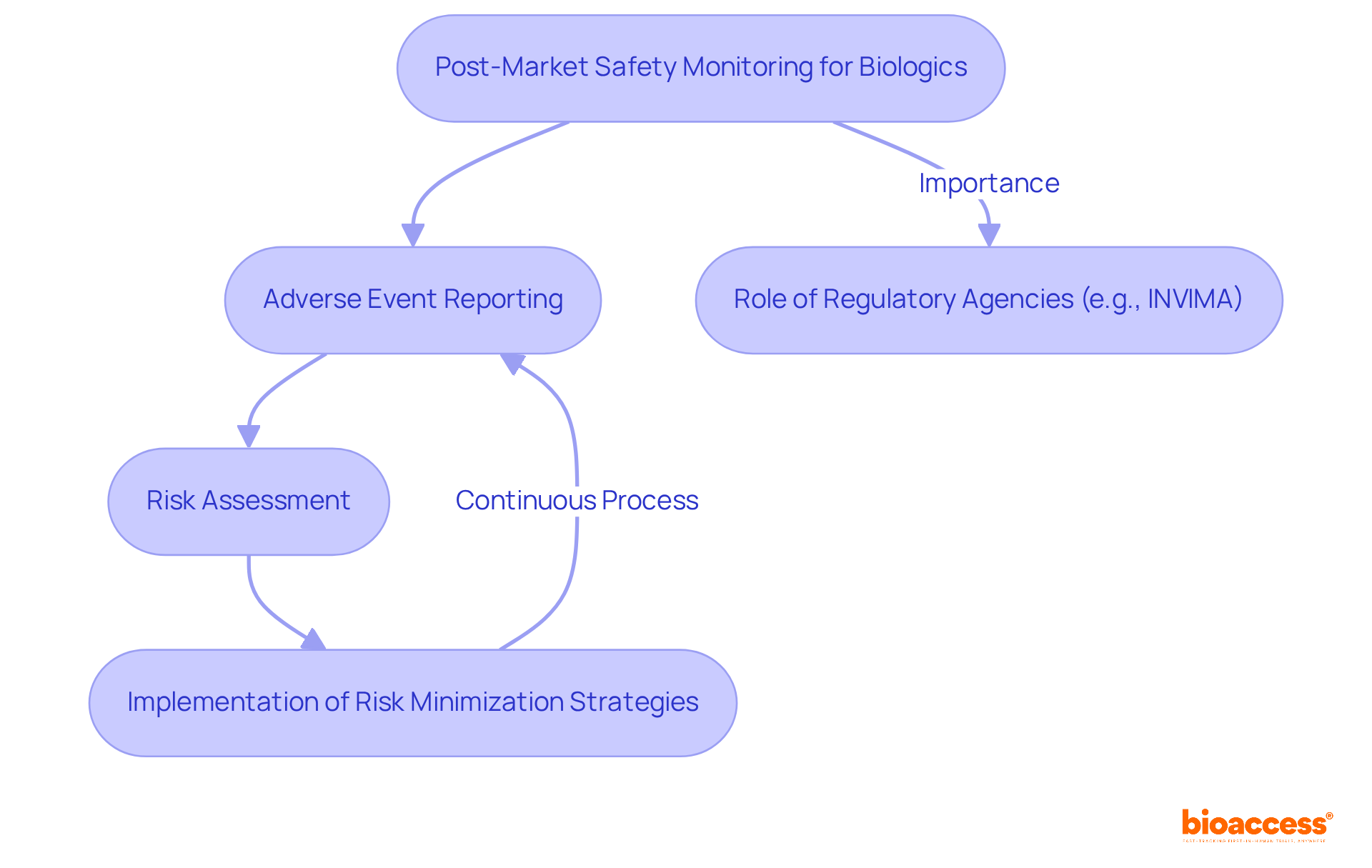
Post-market safety monitoring for biologics involves systematically collecting, analyzing, and interpreting data on the effectiveness and risks of biological products after they receive market approval. This process is essential for identifying adverse effects or unexpected outcomes that may arise once the product is widely used.
What activities are included in post-market safety monitoring?
The activities involved in post-market safety monitoring include adverse event reporting, risk assessment, and the implementation of risk minimization strategies.
Why is post-market safety monitoring important?
It is important because it ensures that any concerns regarding the safety and effectiveness of biologics are swiftly addressed, thereby safeguarding patient well-being and maintaining public trust in these products.
What role do regulatory agencies play in post-market safety monitoring?
Regulatory agencies, such as INVIMA in Colombia, play a vital role in monitoring the quality of health products. They are responsible for inspecting and supervising the marketing and manufacturing of health products, ensuring their safety and efficacy.
What classification does INVIMA hold and why is it significant?
INVIMA is classified as a Level 4 health authority by the Pan American Health Organization/World Health Organization. This classification highlights the importance of robust regulatory frameworks in ensuring the safety and efficacy of biologics.
What challenges exist in post-market safety monitoring for biologics in Bulgaria?
Key challenges include the need for enhanced collaboration among stakeholders to strengthen regulatory practices and improve patient outcomes.
What are the next steps for improving post-market safety monitoring in Bulgaria?
The next steps involve fostering partnerships and sharing insights among stakeholders to enhance post-market safety monitoring and ensure that biologics continue to meet safety and effectiveness standards.