


This article centers on mastering clinical trials and data management to guarantee research success. It outlines the essential phases of clinical studies while emphasizing the significance of effective data handling, adherence to regulatory standards, and innovative recruitment strategies. By detailing how structured oversight, technology integration, and community engagement can enhance trial efficiency and participant trust, the article illustrates the pathway to achieving higher-quality outcomes and reduced recruitment times.
In the context of the Medtech landscape, understanding these dynamics is crucial. The role of bioaccess in addressing key challenges cannot be overstated. As we navigate the complexities of clinical research, it is imperative to consider how collaboration and strategic planning can lead to more effective trials.
Ultimately, the importance of collaboration in clinical research is paramount. Moving forward, stakeholders must prioritize these strategies to foster innovation and improve outcomes. The next steps involve a commitment to enhancing trial processes and ensuring that participant engagement remains at the forefront of research initiatives.
Understanding the intricacies of clinical trials and data management is crucial in the fast-evolving landscape of medical research. The success of new therapies hinges on rigorous testing and precise data handling. Stakeholders must navigate a myriad of challenges, from recruitment hurdles to regulatory compliance. This article delves into best practices that streamline the clinical trial process while enhancing data integrity and participant engagement.
How can organizations leverage innovative strategies to overcome these obstacles and ensure successful outcomes in their research endeavors?
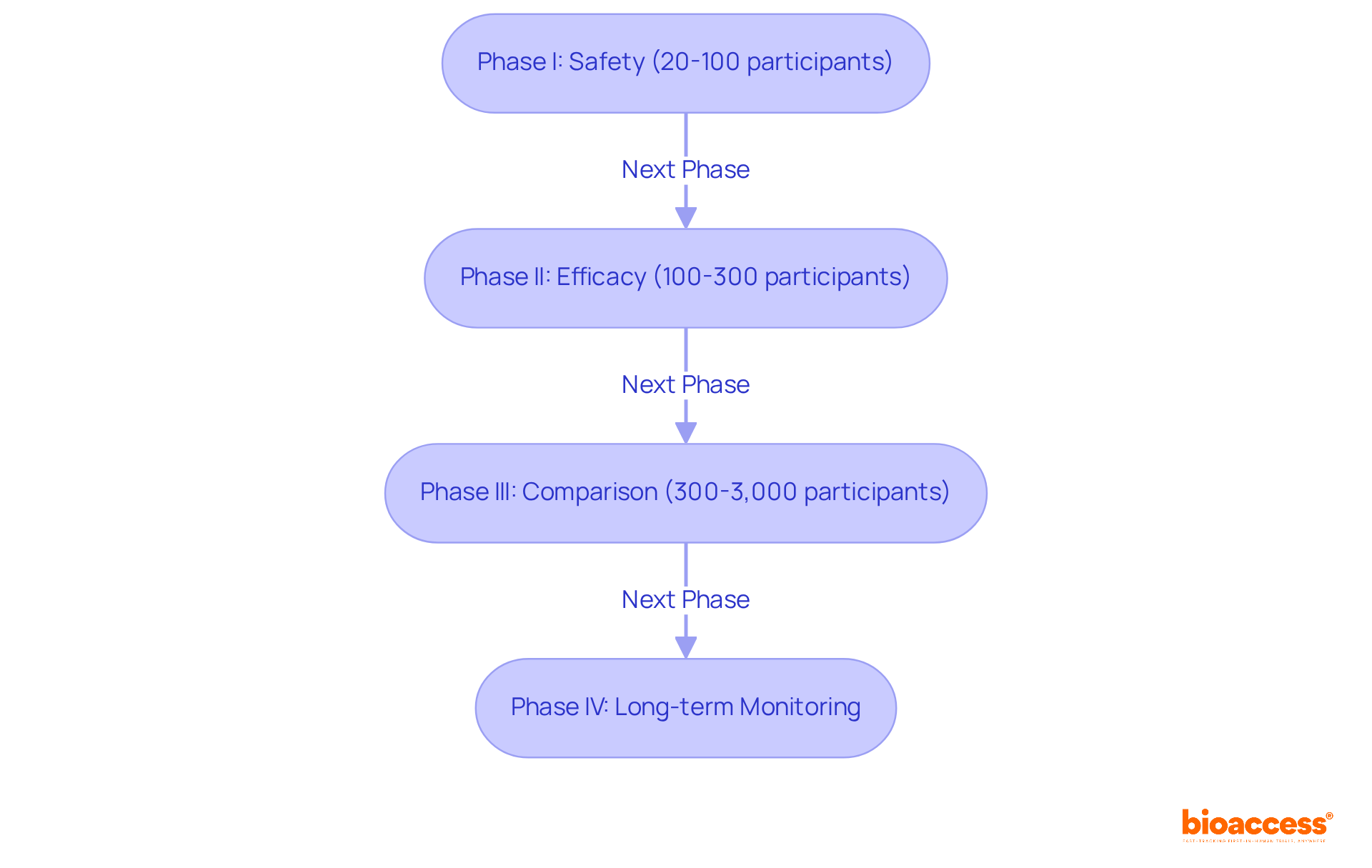
Clinical studies are organized examinations designed to evaluate the safety and effectiveness of new medical treatments. They progress through four distinct phases:
Comprehending these stages is essential for efficient testing oversight, as roughly 5-14% of therapies that enter research studies successfully finish all stages and gain authorization. Furthermore, it is significant that around 80% of medical studies are postponed or terminated because of recruitment issues, highlighting the necessity of efficient information handling and recruitment approaches.
The handling of information in clinical trials and data management involves the gathering, validation, and examination of information to guarantee its integrity and reliability. A strong information oversight strategy (DMP) is crucial for effective clinical trials and data management, detailing processes for information handling and adherence to regulatory standards. The application of electronic information capture (EDC) systems improves efficiency and precision, while compliance with Good Clinical Practice (GCP) guidelines guarantees high-quality information throughout the study process. For example, bioaccess® provides extensive services that encompass feasibility studies, site selection, compliance reviews, setup for experiments, import permits, project oversight, and reporting, which are essential for addressing regulatory obstacles and speeding up the research process. Additionally, bioaccess® links cutting-edge Medtech, Biopharma, and Radiopharma startups with leading research locations, enabling quicker patient enrollment and substantial cost reductions of $25K per patient with FDA-ready information.
In conclusion, efficient information oversight is essential for the success of clinical trials and data management, as it directly influences the dependability of outcomes and the overall effectiveness of the research process. With the growing intricacy of experiments, a strategic method to clinical trials and data management, supported by professional services such as those offered by bioaccess®, is more crucial than ever. Furthermore, the economic influence of research studies, encompassing job creation and healthcare enhancement, emphasizes the wider importance of effective management of experiments.
Adhering to regulatory standards set by agencies such as the FDA and EMA is crucial for the success of clinical studies. Researchers must conduct their studies in accordance with Good Clinical Practice (GCP), which requires:
Ethical frameworks, including the Declaration of Helsinki, delineate responsible research practices. In this context, bioaccess offers a comprehensive suite of services, including:
This ensures that all activities align with regulatory requirements.
Understanding informed consent rates is particularly vital; studies reveal that while 97.5% of participants comprehend confidentiality, only 4.8% grasp the concept of a placebo. Continuous training and updates on regulatory changes are imperative for all team members to ensure adherence and uphold ethical standards throughout the research process. This commitment not only bolsters participant trust but also enhances the integrity and validity of research outcomes. Collaboration in these areas is essential for navigating the complexities of clinical research successfully.

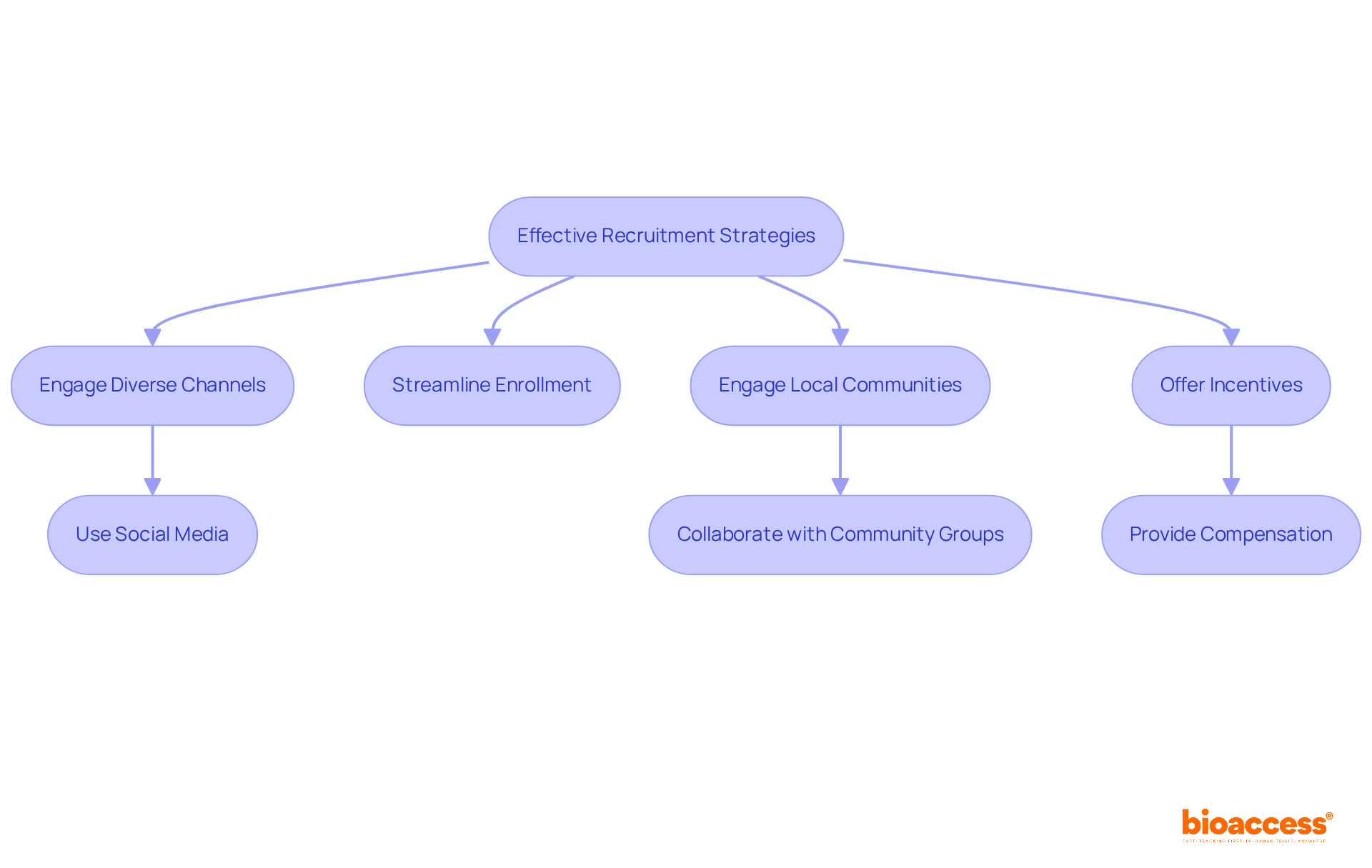
To enhance recruitment for research studies, it is essential to apply a focused approach that aligns with the demographics and traits of the desired participant group. Engaging diverse channels—such as social media, healthcare providers, and patient advocacy groups—can significantly expand outreach efforts. Utilizing social media platforms, for instance, has proven effective in reaching younger demographics, who may be more inclined to participate in research studies. Furthermore, streamlining the enrollment procedure and providing clear, accessible information about the study can alleviate potential participants' concerns, fostering a sense of trust and transparency.
Engaging local communities is vital for improving recruitment rates. This can be achieved through collaborations with community groups and conducting informational sessions that address prevalent misunderstandings about research studies. Additionally, offering incentives for participation, such as compensation for travel or time, can further motivate individuals to enroll. Regularly assessing recruitment strategies and adapting them based on participant feedback and outcomes is crucial for sustained success.
In Colombia, the partnership between bioaccess™ and Caribbean Health Group aims to establish Barranquilla as a leading location for medical studies in Latin America, supported by the Minister of Health. This initiative not only enhances recruitment strategies but also contributes to local economic growth through job creation and improved healthcare services. Notably, the partnership has achieved over a 50% reduction in recruitment time and a 95% retention rate, underscoring the effectiveness of these strategies. As highlighted in recent research, fewer than 4% of adults in the US engage in research studies, emphasizing the urgent need for effective recruitment strategies.
Utilizing technology in clinical trials significantly enhances information management efficiency. The implementation of electronic information capture (EDC) systems facilitates real-time input and monitoring, effectively reducing the mistakes often associated with manual information handling. EDC systems can achieve error rates as low as 0.04%, showcasing their reliability in contrast to traditional methods, where manual record entry can result in errors in up to 30% of entries.
Furthermore, clinical trials and data management streamline project management, tracking, and reporting processes, contributing to operational savings of up to 62% through the automation of information gathering. The integration of advanced analytics and machine learning tools allows for the identification of trends and anomalies in data, enabling quicker and more informed decision-making.
Ensuring that all technology solutions are user-friendly and seamlessly integrated enhances team collaboration and data accuracy, ultimately leading to more successful trial outcomes. As the eClinical solutions market is projected to reach USD 17.61 billion by 2025, the adoption of these technologies is becoming increasingly vital for the success of clinical research.

Mastering clinical trials and data management is pivotal for ensuring the success of medical research. By understanding the intricacies of the clinical trial phases, adhering to regulatory standards, and implementing effective recruitment strategies, researchers can significantly enhance the integrity and reliability of their studies. The integration of advanced technologies further streamlines data management processes, ultimately leading to improved outcomes and efficiency in clinical research.
Key insights from the article highlight the importance of a well-structured approach to clinical trials. The phases of clinical trials—from safety assessments in Phase I to long-term monitoring in Phase IV—underscore the critical nature of each stage in the research process. Additionally, maintaining compliance with ethical guidelines and regulatory standards is essential for fostering participant trust and ensuring the validity of research findings. Recruitment strategies that engage diverse communities and utilize digital platforms are crucial for overcoming common challenges in participant enrollment.
In light of these discussions, the significance of mastering clinical trials and data management cannot be overstated. As the landscape of medical research continues to evolve, embracing innovative strategies and technologies will be vital for driving progress. Researchers, stakeholders, and organizations must prioritize these best practices to not only enhance the quality of clinical studies but also to contribute to the broader goals of improving healthcare outcomes and fostering economic growth in the field.
What are clinical trials?
Clinical trials are organized examinations designed to evaluate the safety and effectiveness of new medical treatments.
What are the phases of clinical trials?
Clinical trials progress through four distinct phases: Phase I focuses on safety with 20-100 healthy volunteers. Phase II evaluates efficacy with 100-300 participants. Phase III compares new treatments against standard therapies, typically involving 300-3,000 participants over 1 to 4 years. Phase IV monitors long-term effects post-approval.
What percentage of therapies successfully complete all phases of clinical trials?
Roughly 5-14% of therapies that enter research studies successfully finish all stages and gain authorization.
Why are many medical studies postponed or terminated?
Around 80% of medical studies are postponed or terminated due to recruitment issues.
What is the importance of data management in clinical trials?
Data management involves gathering, validating, and examining information to ensure its integrity and reliability, which is crucial for the success of clinical trials.
What is a data management plan (DMP)?
A data management plan (DMP) is a strategy that details processes for information handling and adherence to regulatory standards in clinical trials.
How do electronic data capture (EDC) systems benefit clinical trials?
EDC systems improve efficiency and precision in data collection and management during clinical trials.
What guidelines ensure high-quality data in clinical studies?
Compliance with Good Clinical Practice (GCP) guidelines ensures high-quality data throughout the study process.
What services does bioaccess® provide for clinical trials?
Bioaccess® provides services including feasibility studies, site selection, compliance reviews, experiment setup, import permits, project oversight, and reporting, which help address regulatory obstacles and speed up the research process.
How does bioaccess® impact patient enrollment and costs?
Bioaccess® connects Medtech, Biopharma, and Radiopharma startups with leading research locations, enabling quicker patient enrollment and reducing costs by $25K per patient with FDA-ready data.
What is the broader economic impact of effective clinical trial management?
Effective management of clinical trials contributes to job creation and healthcare enhancement, emphasizing its wider importance in the research landscape.