


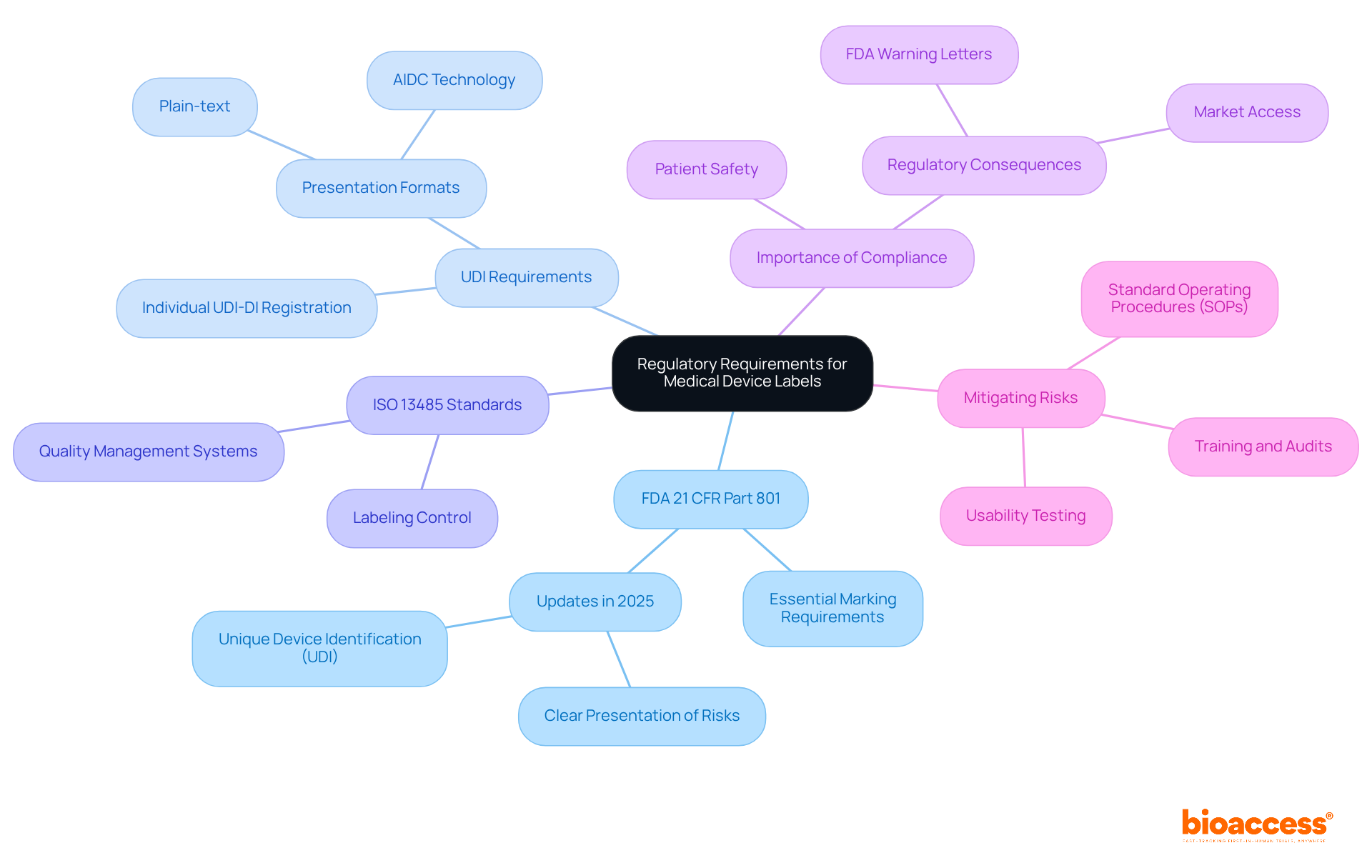
The article addresses essential compliance practices for master labels of medical devices, underscoring the critical nature of adhering to regulatory requirements established by the FDA and global standards to ensure patient safety and product efficacy. It elaborates on this by detailing vital labeling elements, upcoming regulatory updates, and best practices aimed at enhancing clarity and usability. These components are crucial not only for maintaining compliance but also for fostering user trust in medical products.
Navigating the intricate landscape of medical device labeling is critical for manufacturers aiming to ensure compliance and safeguard patient safety. With evolving regulations, such as the anticipated updates to FDA's 21 CFR Part 801 in 2025, the stakes are higher than ever for producers to understand the essential components of effective labeling.
How can manufacturers not only meet these regulatory requirements but also enhance the clarity and usability of their labels to foster trust and reliability among users?
This article delves into key compliance practices that will empower manufacturers to excel in this vital aspect of medical device production.
Navigating the complex terrain of medical product identification requires producers to thoroughly understand the regulatory framework established by the FDA and global standards organizations. Central to this framework is the FDA's 21 CFR Part 801, which delineates essential marking requirements for medical equipment. This includes critical information such as the equipment name, intended use, and manufacturer details. Compliance with these regulations is not merely a formality; it is vital for ensuring patient safety and product efficacy.
In 2025, updates to 21 CFR Part 801 will further refine packaging practices, emphasizing the necessity for clear presentation of risks and precautions. For instance, the introduction of Unique Device Identification (UDI) requirements enhances traceability, facilitating better monitoring of device performance and safety. Additionally, manufacturers must adhere to ISO 13485 standards, which incorporate quality management systems into marking processes, ensuring that all tags are accurate and compliant.
Statistics reveal that identification errors frequently prompt FDA warning letters, underscoring the importance of meticulous labels for medical devices design and validation. Regular training and audits are essential for maintaining compliance and adapting to evolving regulations. By implementing standard operating procedures (SOPs) for the review of labels for medical devices and conducting usability testing, manufacturers can mitigate risks associated with inaccuracies in labels for medical devices, ultimately safeguarding public health and enhancing market access.
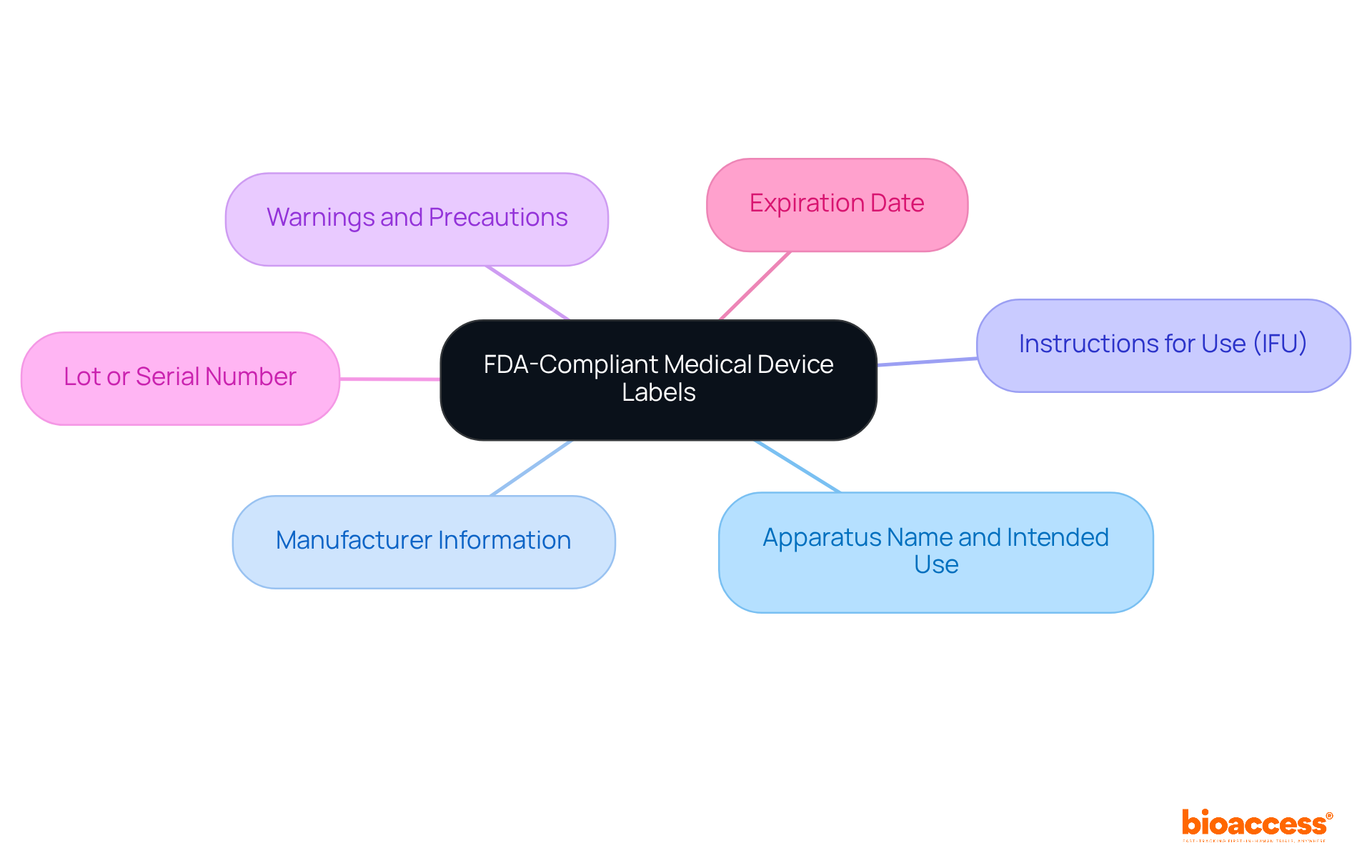
Labels for medical devices that are FDA-compliant must incorporate several essential elements to ensure clarity and safety. These elements include:
By incorporating these elements, producers not only enhance user safety but also ensure compliance with regulatory standards related to labels for medical devices, thereby fostering trust and reliability in their products. Experts such as Ana Criado, Director of Regulatory Affairs and a professor in biomedical engineering, highlight the significance of these factors in navigating the intricate regulatory pathways for medical technologies, especially in markets like Colombia where adherence is essential for effective product launch.
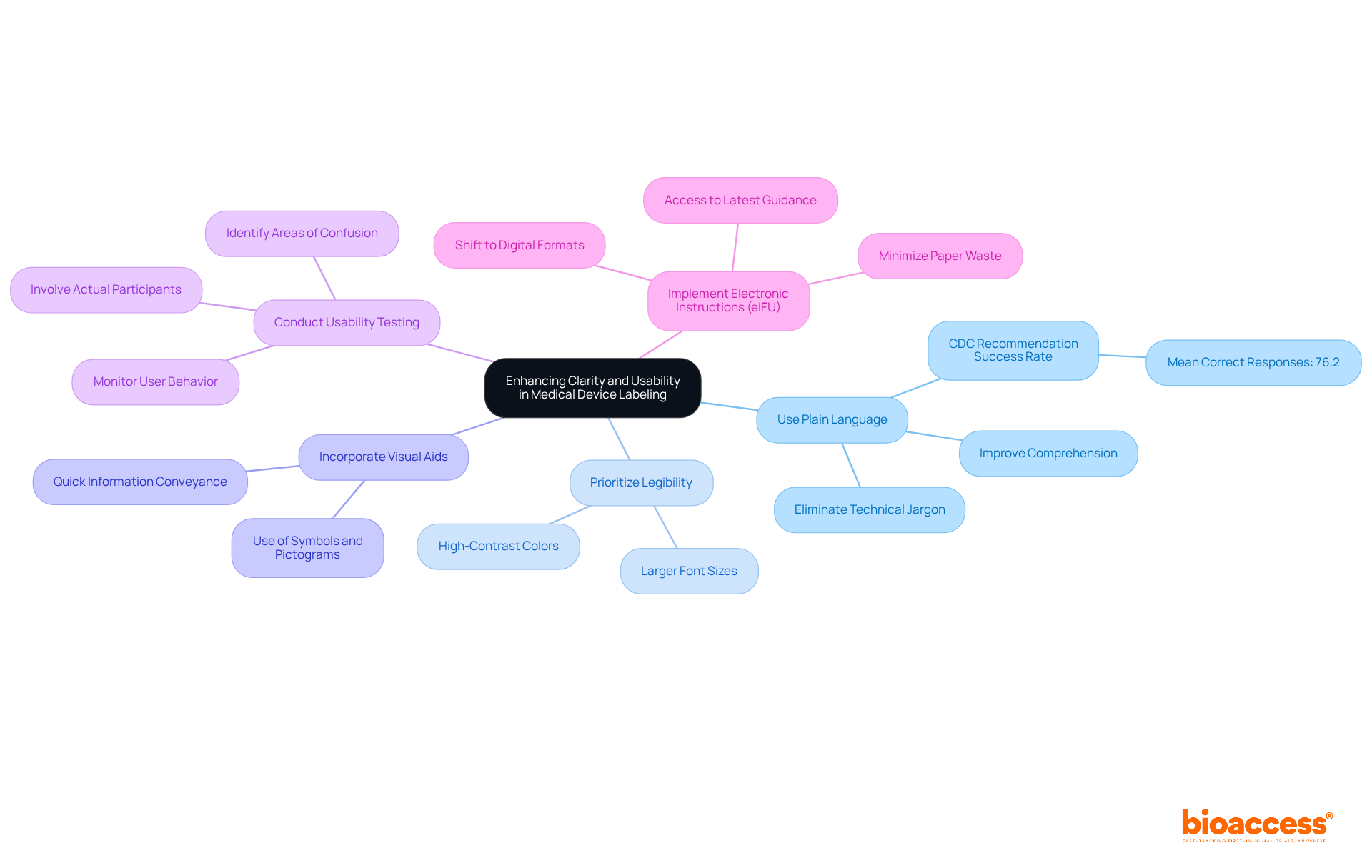
To enhance clarity and usability in medical device labeling, manufacturers must adopt the following best practices:
Use Plain Language: Eliminate technical jargon to ensure that the language is accessible to everyone, enhancing comprehension and reducing the risk of misuse. As Steve Krug states, "If you want a great site, you’ve got to test," emphasizing the importance of clear communication in usability.
Prioritize Legibility: Utilize high-contrast colors and larger font sizes to guarantee that text is easily readable, accommodating individuals with diverse visual abilities.
Incorporate Visual Aids: Utilize symbols and pictograms to convey critical information quickly and effectively, assisting individuals in understanding essential instructions at a glance.
Conduct Usability Testing: Involve actual participants in testing the labels to pinpoint areas of confusion and progressively enhance the design. This approach aligns with the principle that usability drives the acceptance of new technologies. Monitoring individual behavior, as Jakob Nielsen recommends, offers more valuable insights than depending exclusively on feedback from individuals.
Implement Electronic Instructions for Use (eIFU): Shifting to digital formats enables the delivery of current information, minimizing paper waste and ensuring individuals have access to the latest guidance.
By focusing on these strategies, manufacturers can create labels that not only meet regulatory requirements but also significantly enhance user experience and safety. Notably, studies show that using plain language can improve comprehension, with a mean percentage of correct responses for plain language recommendations reaching 76.2% for the CDC recommendation. This highlights the effectiveness of clear communication in medical device labeling.
Navigating the landscape of medical device labeling is crucial for ensuring compliance with regulatory standards and enhancing patient safety. This article underscores the importance of understanding the intricate regulations set forth by the FDA and other global organizations, particularly the upcoming updates to 21 CFR Part 801 in 2025. These regulations dictate essential labeling requirements while emphasizing the need for clear communication of risks and effective product identification through Unique Device Identification (UDI).
Key elements of FDA-compliant labels—such as:
are vital in fostering user safety and trust. By prioritizing clarity and usability through practices like:
manufacturers can significantly improve the effectiveness of their labels. Regular training and usability testing further ensure that labels meet both regulatory requirements and user needs, ultimately safeguarding public health.
As the medical device industry evolves, embracing these compliance practices and focusing on user-centered design will be essential for manufacturers. The commitment to clear, accurate, and accessible labeling not only meets regulatory obligations but also enhances the overall user experience. By prioritizing these elements, stakeholders can contribute to a safer healthcare environment and facilitate better market access, reinforcing the significance of diligent labeling practices in the medical device sector.
What are the key regulatory requirements for medical device labels?
Key regulatory requirements for medical device labels include compliance with the FDA's 21 CFR Part 801, which outlines essential marking requirements such as the equipment name, intended use, and manufacturer details.
Why is compliance with labeling regulations important?
Compliance with labeling regulations is vital for ensuring patient safety and product efficacy, as improper labeling can lead to identification errors and potential safety risks.
What updates to 21 CFR Part 801 are expected in 2025?
The updates to 21 CFR Part 801 in 2025 will refine packaging practices, emphasizing the clear presentation of risks and precautions, and will introduce Unique Device Identification (UDI) requirements to enhance traceability.
What is the role of Unique Device Identification (UDI)?
The UDI enhances traceability of medical devices, facilitating better monitoring of device performance and safety.
What standards must manufacturers adhere to in addition to FDA regulations?
Manufacturers must also adhere to ISO 13485 standards, which incorporate quality management systems into marking processes to ensure accuracy and compliance of labels.
What are the consequences of identification errors in medical device labeling?
Identification errors can prompt FDA warning letters, highlighting the critical importance of meticulous label design and validation.
How can manufacturers maintain compliance with labeling regulations?
Manufacturers can maintain compliance by conducting regular training and audits, implementing standard operating procedures (SOPs) for label review, and performing usability testing to mitigate risks associated with labeling inaccuracies.