


Mastering the MDSAP audit approach requires a clear understanding of the MDSAP framework, thorough preparation through gap assessments, the development of standard operating procedures, and active engagement with auditing organizations. These essential steps are crucial as they ensure compliance with global standards, enhance operational efficiency, and significantly increase the likelihood of favorable audit outcomes. Such preparation strategies are vital for success in the clinical research landscape.
Mastering the Medical Device Single Audit Program (MDSAP) is essential for manufacturers aiming to streamline compliance across multiple regulatory landscapes. This globally recognized initiative simplifies the evaluation process and enhances the credibility and market readiness of medical devices. However, many producers grapple with the complexities of the MDSAP framework. This raises a critical question: what are the essential steps to ensure a successful audit and, ultimately, compliance?
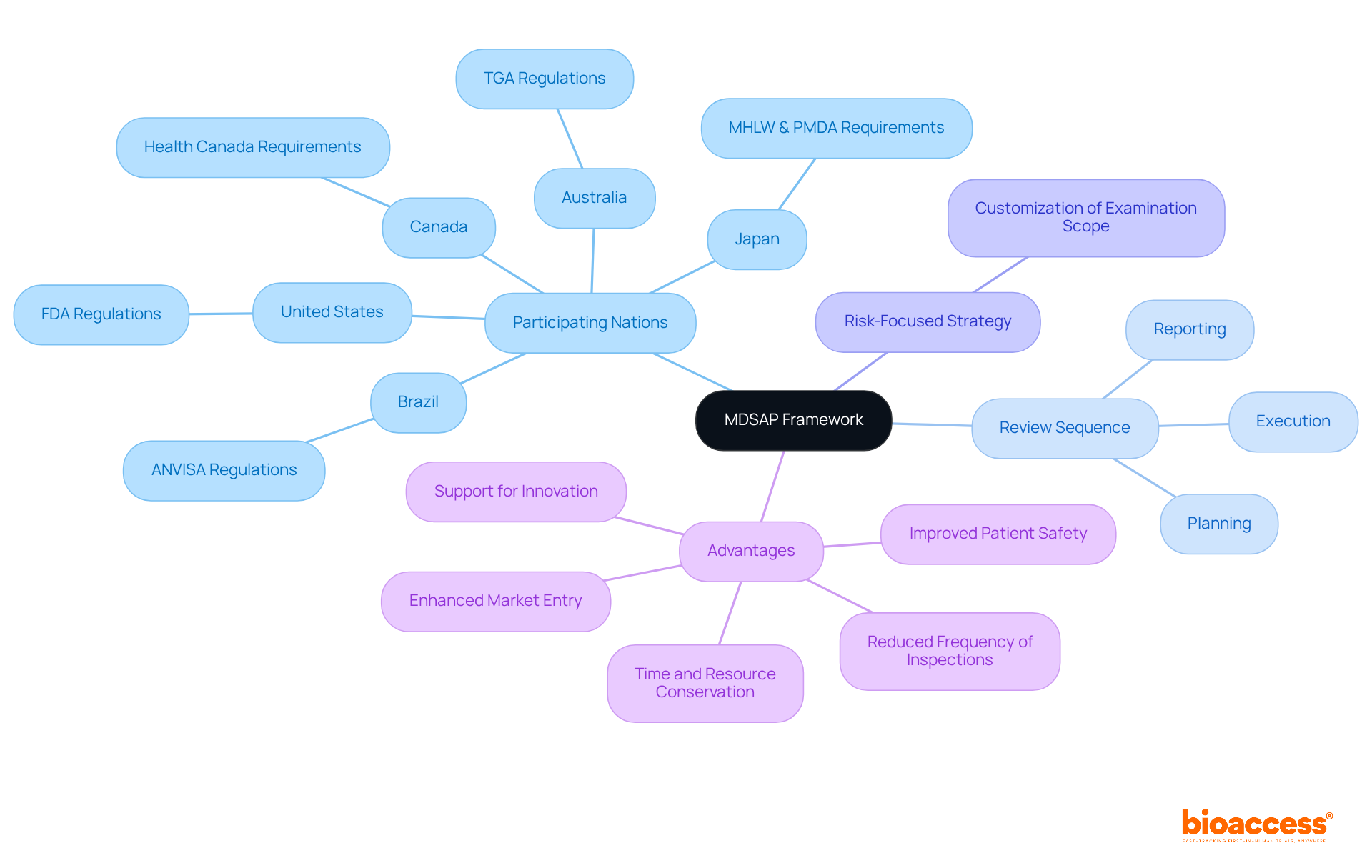
The Medical Device Single Evaluation Program (MDSAP) stands as a pivotal global initiative, enabling a singular regulatory review of a medical device producer's quality management system to meet the diverse needs of regulatory bodies. For producers seeking to streamline compliance and alleviate the burden of multiple evaluations, a thorough understanding of this framework is imperative. The key components of the MDSAP framework encompass:
Understanding these components is vital for effective preparation for the MDSAP audit approach, ensuring compliance with global standards and improving operational efficiency. By comprehending the regulatory landscape shaped by INVIMA, producers can adeptly navigate the intricacies of compliance and leverage insights from experts such as Katherine Ruiz, who specializes in regulatory affairs for medical devices and in vitro diagnostics in Colombia.
To effectively prepare for the MDSAP audit, manufacturers should implement the following key steps:
Conduct a Gap Assessment: Begin by evaluating your current quality management system (QMS) against the applicable requirements. This assessment is crucial for identifying compliance gaps and formulating an action plan to address them. Regular gap analyses have proven essential in the medical device sector, as they help organizations pinpoint weaknesses and enhance overall quality. Dr. Fiona Masterson stresses that gap assessments are 'essential tools' in this procedure.
Develop Standard Operating Procedures (SOPs): Ensure that all activities are documented in SOPs that align with MDSAP standards. This documentation should cover all elements of the QMS, including design, production, and post-market surveillance, to enable a seamless review.
Train Your Team: Conduct comprehensive training sessions for all staff engaged in the evaluation process. It is essential that they comprehend MDSAP criteria and their distinct responsibilities in upholding compliance, as well-prepared groups greatly aid in achieving favorable review results.
Organize Documentation: Collect all essential documentation, including records of past evaluations, corrective measures implemented, and any modifications made to the QMS. Making certain that these documents are readily available for inspectors can simplify the review and lessen possible delays.
Conduct Internal Evaluations: Simulate the medical device single assessment program process through internal evaluations. This proactive method aids in recognizing any outstanding problems related to the MDSAP audit approach, enabling corrective measures to be taken prior to the formal review, thus improving overall preparedness. Carrying out an internal review is suggested to align with the MDSAP audit approach.
Engage with Auditing Organizations: Establish communication with recognized auditing organizations (AOs) to understand their specific requirements and expectations. This engagement can offer valuable insights into the evaluation process and assist in customizing your preparation efforts accordingly.
Establish a Corrective Action Plan: Prepare a detailed strategy for addressing any nonconformities identified during internal reviews. This plan should outline steps for resolution and timelines for implementation, ensuring that all issues are systematically addressed.
By diligently following these steps, producers can significantly improve their preparedness for the assessment, ultimately boosting their likelihood of attaining a favorable result. It is important to note that the evaluation can take between 6 to 12 months to arrange due to high demand for assessors. Regular assessments of QMS implementation are crucial to avoid non-compliance, which can lead to delays in product market entry and financial repercussions.

To effectively navigate the MDSAP audit, manufacturers can leverage a variety of resources and strategies that are essential for success:
MDSAP Audit Checklist: Develop a comprehensive checklist that encompasses all MDSAP requirements along with the necessary documentation. This tool serves as a roadmap throughout the preparation phase, ensuring that no critical elements are overlooked. It is essential for identifying gaps in a manufacturer’s Quality Management System (QMS), as highlighted by industry experts.
Training Materials: Utilize training resources and webinars provided by recognized auditing organizations. These materials offer valuable insights into the evaluation process and emphasize best practices for achieving compliance, particularly the significance of risk management procedures.
Networking Opportunities: Connect with industry colleagues by participating in forums or workshops focused on the program. Networking can yield valuable insights and shared experiences that enhance comprehension of the evaluation landscape.
Consulting Services: Consider recruiting advisors who specialize in preparation for the medical device single audit program. Their expertise can assist in pinpointing potential obstacles, particularly for smaller producers, and streamline the review process, ultimately resulting in a more efficient experience.
Continuous Monitoring: Establish a system for ongoing compliance monitoring with MDSAP requirements. Routine evaluations and modifications to your QMS are vital for ensuring adherence to standards, as a well-organized QMS is key for successful assessments.
Feedback Mechanisms: Create avenues for collecting feedback from staff involved in the evaluation process. This input can highlight areas requiring enhancement and strengthen overall preparedness.
Post-Assessment Review: Following the assessment, conduct a thorough evaluation of the findings and develop a strategy to address any identified concerns. This proactive approach not only enhances future evaluation performance but also fosters a culture of continuous improvement.
By implementing these strategies, manufacturers can significantly enhance their readiness for the evaluation, navigating the process with greater confidence and efficiency. Furthermore, the flexibility of the MDSAP audit approach, including the option to modify the sequence of audited processes, empowers manufacturers to tailor their approach according to specific needs.

The MDSAP audit approach serves as a pivotal pathway for medical device manufacturers who seek to streamline compliance and bolster their global market presence. By grasping the MDSAP framework and its importance, producers can adeptly prepare for audits that not only fulfill regulatory requirements but also enhance operational efficiencies across diverse markets.
Successful MDSAP audit preparation hinges on several key steps:
Collaborating with auditing organizations and formulating corrective action plans further reinforces the foundation for a favorable evaluation. Leveraging resources such as checklists, training materials, and networking opportunities can significantly enhance readiness and instill confidence throughout the audit process.
Ultimately, embracing the MDSAP audit approach transcends mere compliance; it represents an opportunity for continuous improvement and innovation within the medical device sector. By prioritizing meticulous preparation and utilizing available resources, manufacturers can adeptly navigate the complexities of the audit landscape, ensuring their products not only satisfy regulatory standards but also contribute to enhanced patient safety and care. Proactive steps taken today will pave the way for a prosperous tomorrow in the global marketplace.
What is the MDSAP framework?
The Medical Device Single Evaluation Program (MDSAP) is a global initiative that allows a single regulatory review of a medical device producer's quality management system to meet the requirements of various regulatory bodies.
Which countries participate in the MDSAP program?
The MDSAP program is endorsed by regulatory authorities in the United States, Canada, Brazil, Japan, and Australia.
What is the significance of understanding the MDSAP framework for producers?
Understanding the MDSAP framework is crucial for producers as it helps streamline compliance, reduces the burden of multiple evaluations, and enhances operational efficiency.
What are the key components of the MDSAP framework?
The key components include participating nations, a structured review sequence, and a risk-focused strategy that customizes the examination scope based on specific product offerings and associated risks.
How does the review sequence in MDSAP work?
The review sequence involves planning, execution, and reporting, ensuring a comprehensive evaluation that aligns with ISO 13485 standards.
What advantages does participating in the MDSAP program offer to producers?
Participating in MDSAP allows producers to minimize inspection frequency, save time and resources, enhance their reputation globally, accelerate market entry, foster innovation, and improve patient safety.
How can producers prepare for the MDSAP audit approach?
Effective preparation for the MDSAP audit involves understanding the components of the framework and ensuring compliance with global standards.
Who can provide insights on navigating compliance in the context of MDSAP?
Experts such as Katherine Ruiz, who specializes in regulatory affairs for medical devices and in vitro diagnostics in Colombia, can provide valuable insights on navigating compliance.