


The primary objective of this article is to elucidate the concept and importance of sequential groups in clinical research, specifically their role in facilitating interim analyses and enhancing study efficiency. This discussion is underpinned by an exploration of how sequential group designs enable flexible sample sizes and prompt decision-making based on accumulating data. Such advantages not only improve ethical standards and patient safety but also expedite the development of new medical interventions, thereby underscoring the critical nature of these methodologies in advancing clinical research.
In the realm of clinical research, the methodology employed can significantly impact the outcomes and ethical considerations of medical studies. Sequential groups represent a dynamic framework that allows for interim analyses, revolutionizing how researchers approach data evaluation and decision-making. This article delves into the advantages of sequential group designs, exploring how they not only enhance the efficiency of studies but also prioritize patient safety by facilitating timely decisions on treatment effectiveness.
Yet, despite their growing popularity, what challenges do researchers face in implementing these innovative methodologies? How can they overcome these challenges to optimize research outcomes?
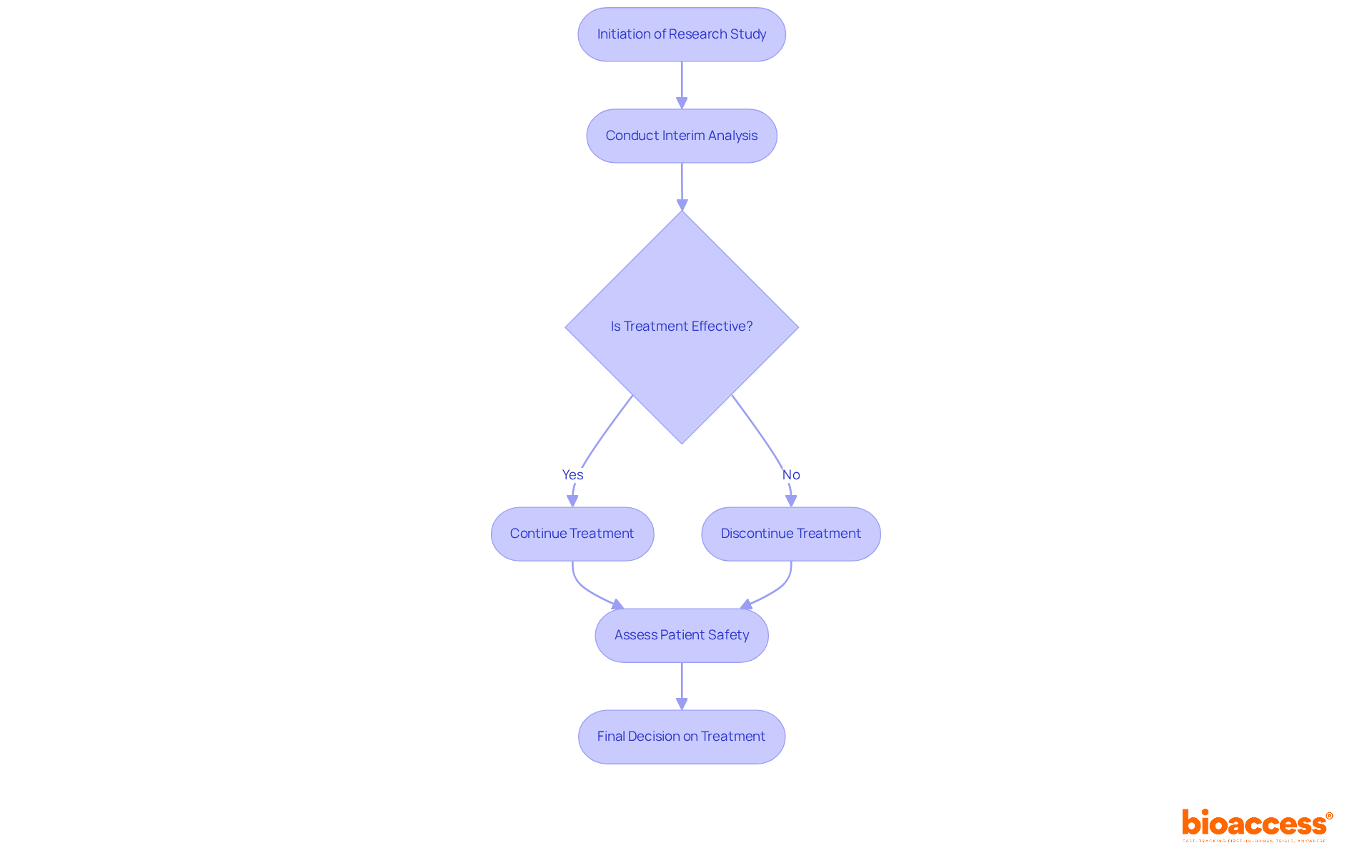
In medical research, a sequential group serves as a framework that facilitates the analysis of data at multiple stages throughout a medical study. This methodology empowers researchers to make provisional decisions informed by the accumulating data, potentially leading to the early termination of the study based on effectiveness or futility.
In adaptive medical studies, the use of a sequential group design proves particularly advantageous, as the sample size is not predetermined, allowing for flexibility according to initial results. This approach enhances the overall effectiveness of medical studies by potentially reducing the number of individuals exposed to ineffective therapies.
Grasping the dynamics of information accrual during recruitment and follow-up is essential for effectively planning interim analyses. Furthermore, the correlation between early findings and final outcomes significantly impacts the efficacy of medical studies.
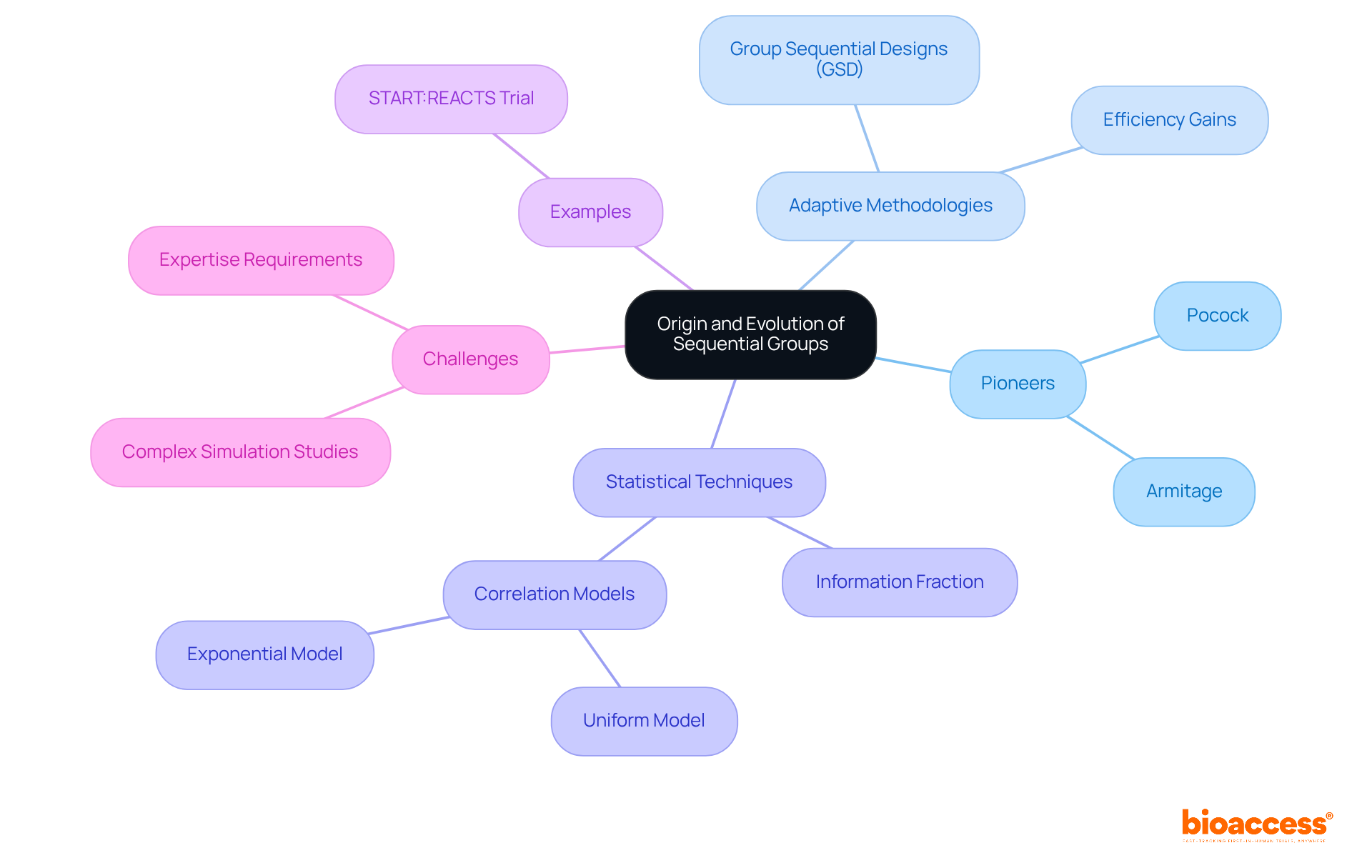
For instance, in the START:REACTS study, the use of a sequential group design facilitated timely decisions based on interim analyses, resulting in the study being discontinued for futility when the observed correlations exceeded expectations. This exemplifies the tangible benefits of this methodology, as it bolsters decision-making processes and ultimately fosters more ethical and effective research outcomes.

A sequential group plays a vital role in enhancing research efficiency and addressing ethical concerns. By enabling interim analyses, these frameworks allow researchers to swiftly identify effective treatments and discontinue those that are ineffective, thereby minimizing patient exposure to suboptimal therapies. This approach not only accelerates the development of new medical interventions but also aligns with ethical standards centered on patient safety.
For instance, in a medical study comparing blinatumomab to standard chemotherapy for children with B-cell acute lymphoblastic leukemia, the study was concluded prematurely after interim analysis revealed significant improvements in event-free survival for the experimental group. Such timely decisions exemplify how a sequential group of approaches can lead to considerable cost savings and reduced timelines for bringing new therapies to market, making them increasingly preferred in the pharmaceutical and biopharmaceutical sectors.
Furthermore, the collaboration between bioaccess™ and Caribbean Health Group, supported by Colombia's Minister of Health, aims to position Barranquilla as a leading site for research trials in Latin America. This partnership enhances recruitment efficiency, achieving over a 50% reduction in recruitment time and impressive retention rates of 95%. This synergy underscores the importance of sequential groups and illustrates how strategic collaborations can significantly improve research outcomes.
The concept of successive sets in health-related research emerged in the mid-20th century as a response to the limitations of traditional fixed-sample approaches. Early pioneers, such as Armitage and Pocock, recognized the need for more flexible study designs that could adapt based on interim results. Their advocacy for adaptive methodologies laid the groundwork for the evolution of sequential group designs (GSD), which are now among the most widely employed methods for adaptive strategies in randomized clinical trials, as noted by expert Nick R. Parsons.
Over the years, improvements in statistical techniques and computational capabilities have facilitated the widespread adoption of frameworks for sequential group analysis. For instance, the information portion at the first interim analysis is often calculated as tau0(t1) = 0.15, illustrating the efficiency gains these approaches can offer compared to fixed structures. The START:REACTS trial serves as a prime example of the practical implementation of GSD, demonstrating how they can enhance patient welfare and optimize resource allocation.
Despite their advantages, challenges to the broader application of sequential approaches remain, particularly the complexity of simulation studies required during the planning phase. These hurdles necessitate substantial expertise and programming skills, which may deter researchers from utilizing these innovative methodologies. Nevertheless, the movement towards more dynamic and responsive research frameworks continues to prioritize patient welfare and the efficient use of resources.
Sequential group designs, which are pivotal in clinical research, are characterized by their capacity for interim analyses that empower researchers to evaluate data at various stages throughout a study. This flexibility in sample size is crucial, allowing for adjustments based on ongoing results, thus optimizing resource utilization and enhancing efficiency.
Early stopping rules, delineated by statistical boundaries for efficacy and futility, inform critical decisions regarding whether to continue, modify, or terminate the study. Such a structured methodology not only adheres to ethical standards in research but also bolsters the reliability and applicability of findings, ultimately fostering more robust practices.
For example, the implementation of interim analyses can reveal significant treatment effects or their absence, prompting timely modifications that align with predefined criteria, ensuring that studies remain relevant and scientifically valid.
At bioaccess®, we leverage over 20 years of expertise in managing research studies, including:
Our comprehensive clinical trial management services guarantee that each phase of the study is meticulously planned and executed, in accordance with the principles of sequential group designs to optimize outcomes and ensure regulatory compliance.

The exploration of sequential groups in clinical research reveals a transformative approach that significantly enhances the efficiency and ethical standards of medical studies. By allowing for interim analyses, this methodology provides researchers with the flexibility to adapt their strategies based on real-time data, ultimately leading to more informed decision-making and improved patient outcomes.
Key insights from the article highlight the historical evolution of sequential group designs, their critical role in optimizing resource utilization, and their capacity to minimize patient exposure to ineffective treatments. Notable examples, such as the START:REACTS study and the collaboration between bioaccess™ and Caribbean Health Group, illustrate the tangible benefits of implementing these frameworks, reinforcing their significance in advancing medical research.
As the landscape of clinical research continues to evolve, embracing sequential group methodologies can pave the way for more ethical and effective trials. Researchers are encouraged to consider these adaptive designs not only for their potential to enhance study outcomes but also for their commitment to prioritizing patient welfare and expediting the development of innovative therapies. The future of clinical research hinges on the ability to harness such dynamic frameworks, ensuring that advancements in medicine are both timely and impactful.
What is a sequential group in clinical research?
A sequential group in clinical research is a framework that allows for the analysis of data at multiple stages throughout a medical study, enabling researchers to make provisional decisions based on accumulating data.
How does a sequential group design affect the study's sample size?
In a sequential group design, the sample size is not predetermined, allowing for flexibility in response to initial results, which can enhance the effectiveness of medical studies.
What are the benefits of using a sequential group design in adaptive medical studies?
The use of a sequential group design can reduce the number of individuals exposed to ineffective therapies and allows for early termination of the study based on effectiveness or futility.
Why is understanding information accrual important in sequential group studies?
Understanding the dynamics of information accrual during recruitment and follow-up is essential for effectively planning interim analyses, which can significantly impact the study's outcomes.
Can you provide an example of a study that utilized a sequential group design?
The START:REACTS study is an example where a sequential group design facilitated timely decisions based on interim analyses, leading to the study's discontinuation for futility when observed correlations exceeded expectations.
What is the overall impact of using a sequential group design on medical research?
A sequential group design bolsters decision-making processes and fosters more ethical and effective research outcomes by allowing researchers to make informed decisions based on early findings.