


The article focuses on the career path of clinical research associates (CRAs), clearly detailing the role's responsibilities, educational requirements, essential skills, and opportunities for advancement. It emphasizes that CRAs are crucial for ensuring the integrity and compliance of clinical studies.
With a growing demand for their expertise in the expanding clinical trial market, which is projected to exceed $80 billion by 2025, the importance of this profession cannot be overstated.
The role of a Clinical Research Associate (CRA) is increasingly recognized as a cornerstone of the medical research landscape, effectively bridging the gap between innovative therapies and regulatory compliance. As the clinical trial market is projected to exceed $80 billion by 2025, aspiring CRAs face a unique opportunity to enter a field that not only promises job security but also offers the chance to make a profound impact on patient care. However, with the rapid evolution of technology and the complexities of regulatory challenges, how can one navigate this dynamic career path effectively and ensure ongoing professional growth?
A Clinical Research Associate (CRA) is pivotal in the research process, tasked with overseeing studies and ensuring adherence to regulatory standards and research protocols. Acting as a vital intermediary between study sponsors and research sites, CRAs meticulously supervise the accuracy of data collected during studies. Their responsibilities include:
This oversight is crucial for maintaining the quality and reliability of medical research, which directly impacts patient outcomes and the approval of new therapies.
With the clinical trial market projected to exceed $80 billion by 2025, the demand for skilled Clinical Research Associates is on the rise, underscoring their significance in advancing medical knowledge and enhancing patient care. Experts assert that CRAs are not merely data monitors; they play a fundamental role in fostering compliance and upholding ethical standards throughout the research process. Rajneesh Patil highlights the necessity for CRAs to embrace emerging technologies and analytics tools, showcasing the evolving nature of their responsibilities.
Furthermore, research organizations face challenges such as:
These efforts ultimately contribute to the successful development of innovative therapies, making CRAs indispensable within the research landscape. As the industry increasingly emphasizes diversity in clinical studies, Clinical Research Associates are essential in promoting inclusive study experiences, ensuring that research reflects the diverse populations it aims to serve.
Many Research Associates begin their careers as Research Coordinators or Trial Assistants, providing a pathway for professional growth within the field. The comprehensive services offered by bioaccess—including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting—are critical elements that Clinical Research Associates must navigate to secure successful trial outcomes.
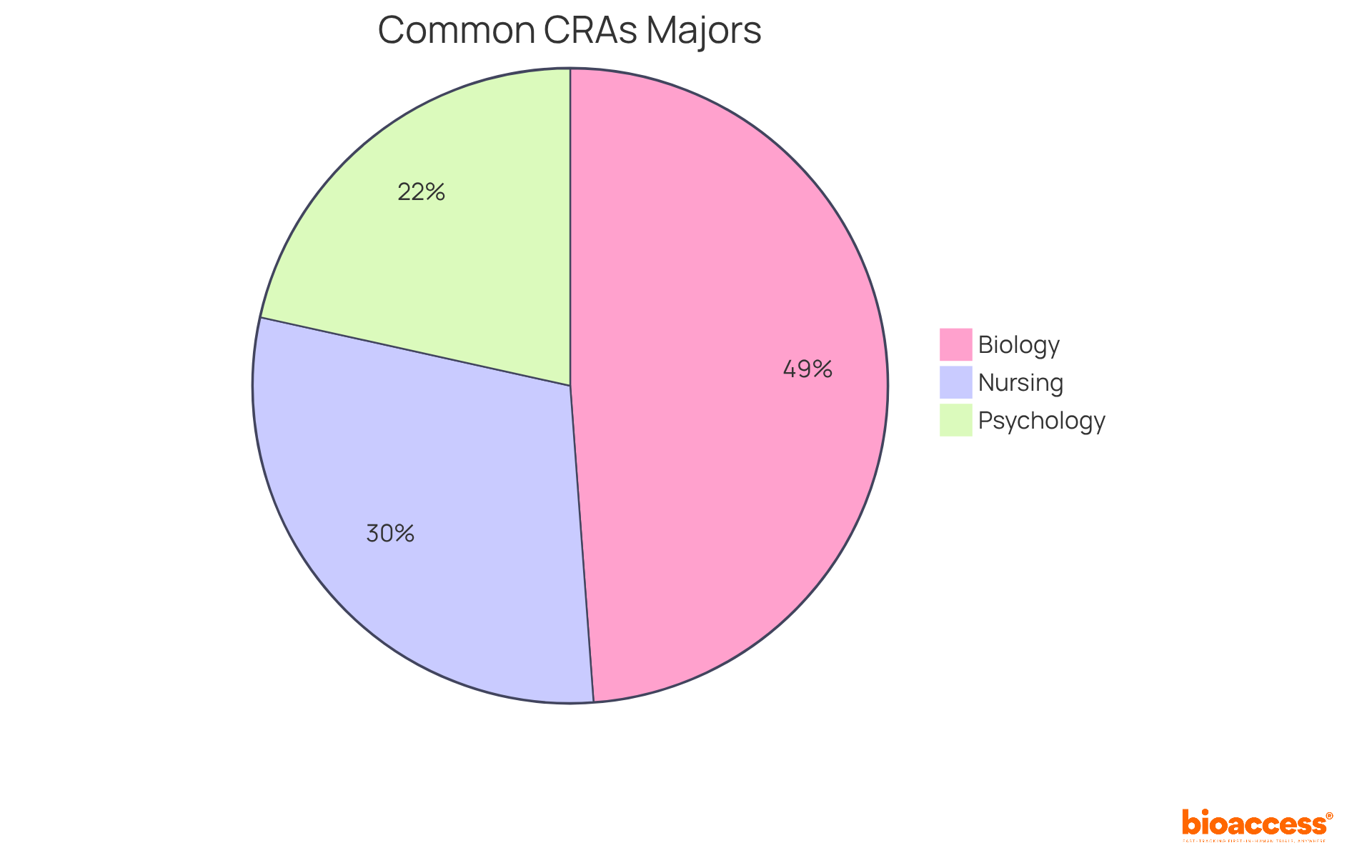
To embark on a clinical research associate career, candidates typically require a bachelor's degree in life sciences, nursing, or a related field. The most prevalent majors for CRAs encompass:
Employers often favor applicants with pertinent clinical study experience, which can be obtained through internships or entry-level roles in clinical research. Certifications such as the Certified Clinical Research Associate (CCRA) from the Association of Clinical Research Professionals (ACRP) or the Clinical Research Associate Certification (CRAC) are increasingly acknowledged as valuable assets, significantly enhancing job prospects and opportunities for career advancement.
In 2025, projections indicate that certified professionals can earn 20-30% more than their uncertified peers, underscoring the critical nature of acquiring these credentials. Furthermore, the median salary for clinical research associates is reported at $100,890 per year, emphasizing the financial advantages of certification. The demand for a clinical research associate career is anticipated to grow by 10% annually, signaling robust job security and opportunities within the field. Continuous education and training in Good Clinical Practice (GCP) are essential for ensuring compliance with industry standards and maintaining a competitive edge in the evolving research landscape.
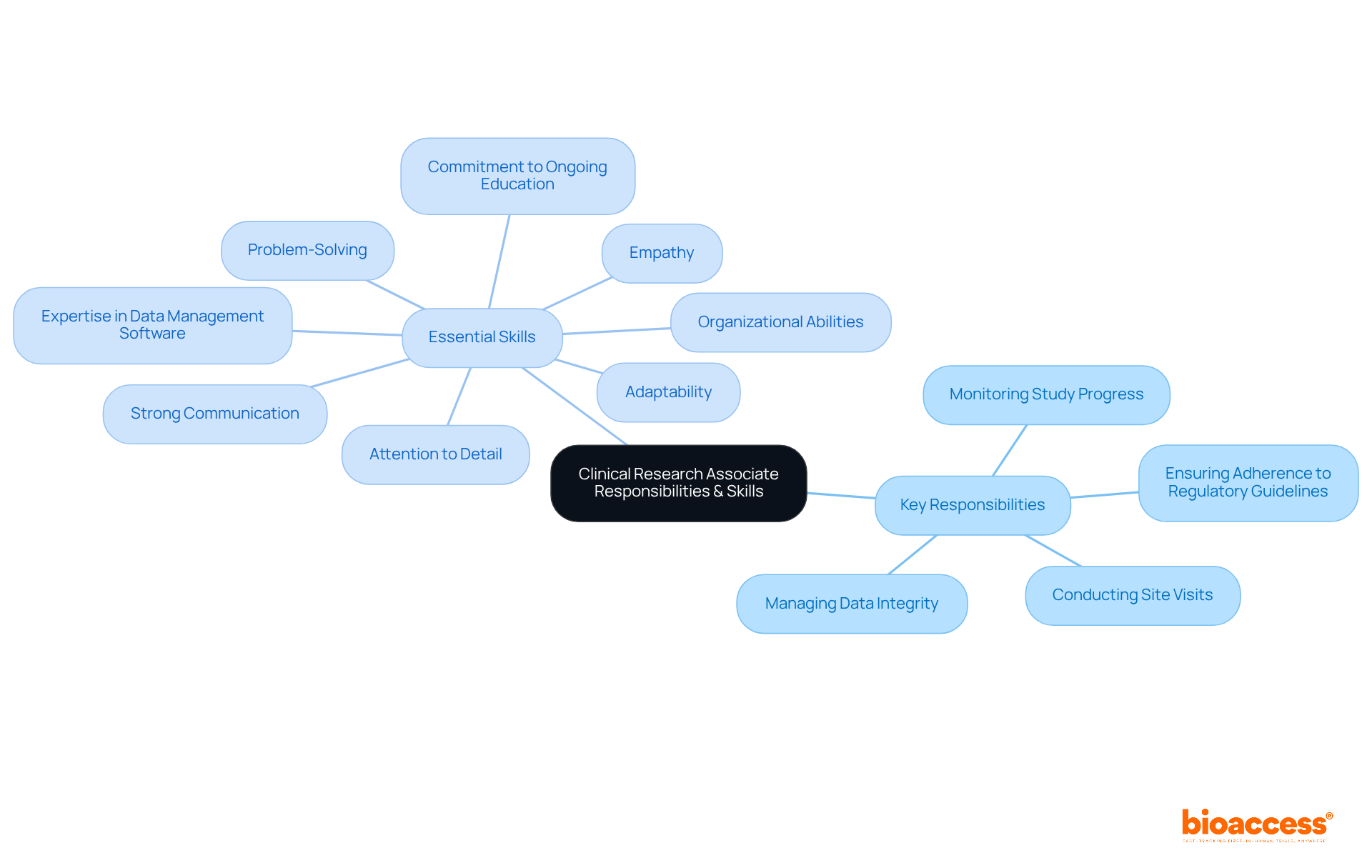
A clinical research associate career is pivotal for the success of research studies. Their key responsibilities encompass:
Backed by extensive clinical study management services offered by bioaccess, which include:
CRAs are essential to the research process. To excel in this dynamic environment, those pursuing a clinical research associate career must possess a diverse skill set. Strong communication skills are vital for effective collaboration with various stakeholders, including investigators, sponsors, and regulatory bodies. Attention to detail is paramount, as even minor oversights can significantly impact study outcomes. Problem-solving skills enable CRAs to navigate obstacles that arise during studies, while organizational abilities help them manage multiple tasks effectively.
As the responsibilities of CRAs evolve, expertise in data management software is increasingly essential. CRAs are tasked with maintaining precise and thorough study data, necessitating a strong comprehension of research study protocols to uphold the integrity of the research process. Industry surveys underscore that adaptability and a commitment to ongoing education are critical qualities for a successful clinical research associate career, given the ever-changing landscape of research. Furthermore, specialists highlight that the ability to listen to patient feedback and integrate it into research procedures can greatly enhance the quality of research results, underscoring the significance of empathy and understanding in this role. Moreover, the impact of Medtech clinical studies extends beyond individual experiments, contributing to job creation, economic development, healthcare improvement, and fostering international collaboration.
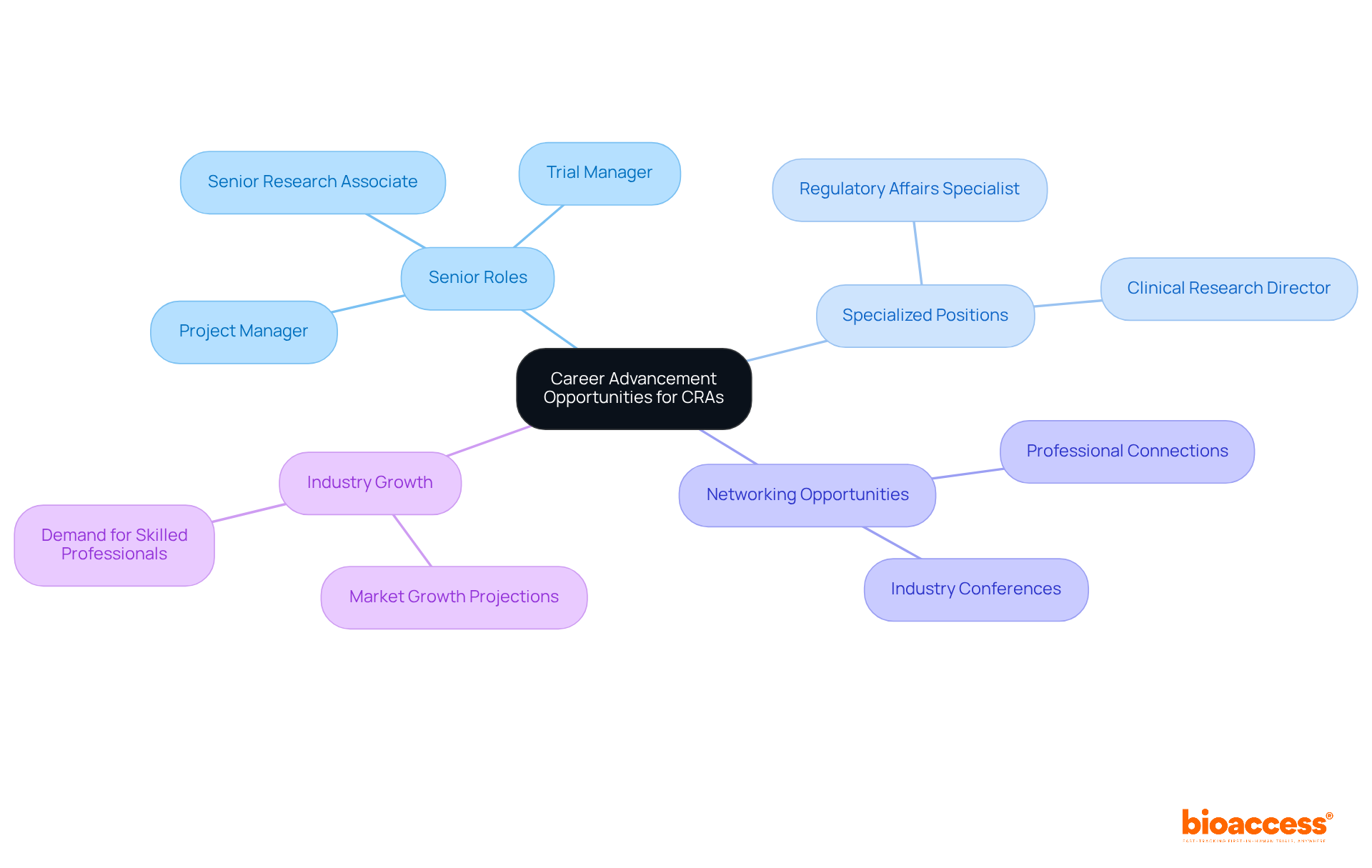
There are abundant career advancement opportunities within a clinical research associate career, with many professionals successfully transitioning to senior research associate roles, trial managers, or project managers. Typically, those pursuing a clinical research associate career can anticipate advancing to senior roles within 5 to 7 years, contingent upon their experience and the complexity of the trials they manage. With further education and training, those pursuing a clinical research associate career may also move into specialized positions such as Regulatory Affairs Specialists or Clinical Research Directors, thereby broadening their career prospects.
Networking and participation in industry conferences are vital in enhancing career prospects, as these activities cultivate connections that can lead to new opportunities. The clinical research sector is witnessing a significant demand for skilled professionals, with the market projected to exceed $80 billion by 2025. This growth translates into a stable and rewarding clinical research associate career path, as these professionals are increasingly recognized for their contributions to advancing medical knowledge and improving patient outcomes.
Numerous success stories exist, with many Clinical Research Associates highlighting their journeys of transitioning to Clinical Trial Managers, which underscores the significance of mentorship and ongoing education in advancing a clinical research associate career. As the industry evolves, it is crucial for credit rating agencies to remain informed about trends and advancements, ensuring they stay competitive in this dynamic field. Insights from successful CRAs often emphasize the significance of perseverance and adaptability, reinforcing the notion that career progression is a marathon, not a sprint.
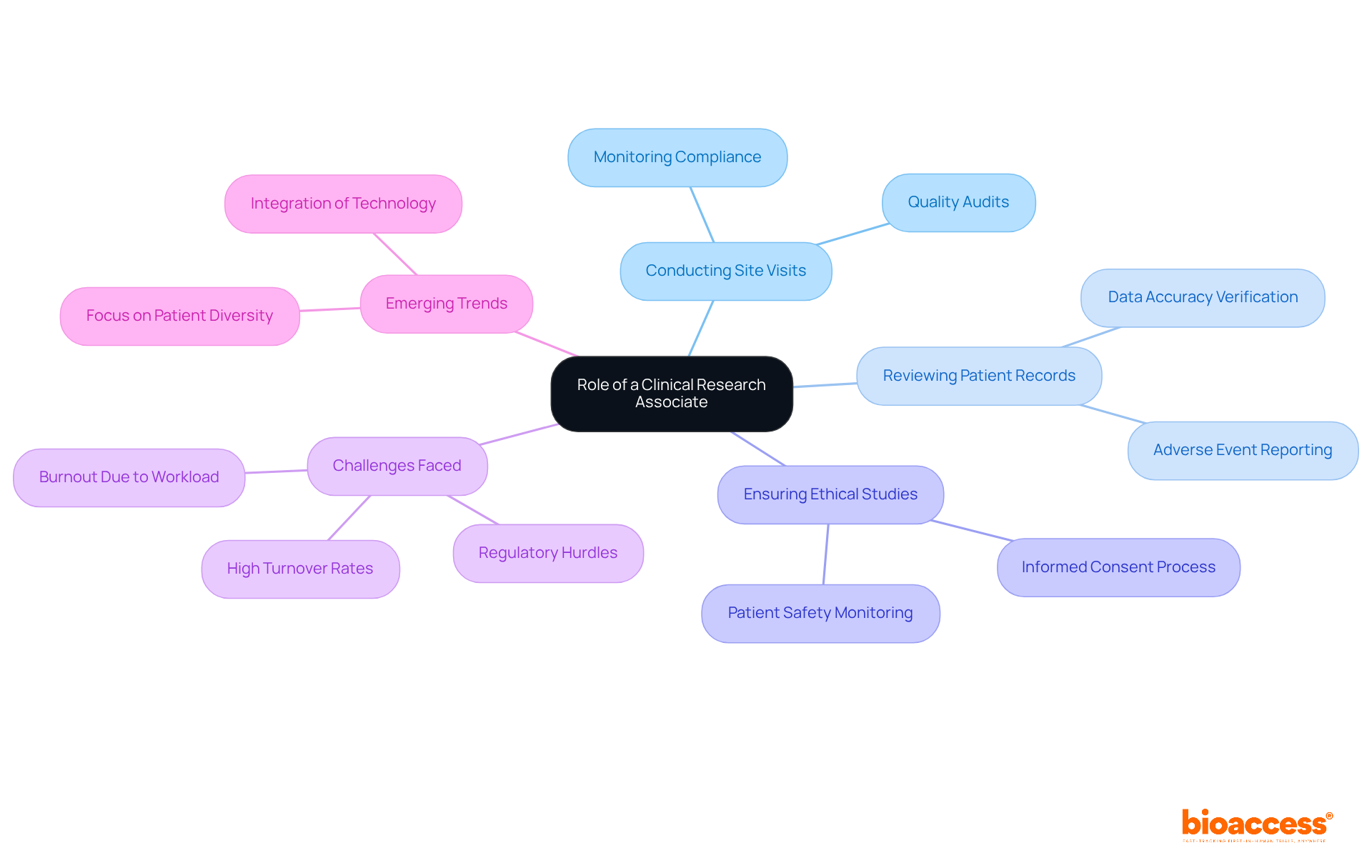
The role of a Clinical Research Associate (CRA) is vital in the realm of clinical trials, ensuring ethical practices and regulatory compliance. As key intermediaries, CRAs oversee the accuracy of data collection, which directly influences patient outcomes and the development of new therapies.
This article underscores the increasing demand for CRAs, fueled by a clinical trial market projected to surpass $80 billion by 2025. Key points include:
As the field continues to evolve, CRAs must prioritize ongoing education and networking to advance their careers. Numerous growth opportunities exist, including transitions to senior roles or specialized positions in regulatory affairs and project management.
In summary, the CRA profession is not only essential for the integrity of clinical research but also plays a significant role in advancing healthcare. By embracing the challenges and responsibilities of this career path, CRAs can enhance their professional prospects while contributing to the future of medical research and patient care.
What is the role of a Clinical Research Associate (CRA)?
A Clinical Research Associate (CRA) oversees clinical studies, ensuring adherence to regulatory standards and research protocols. They act as intermediaries between study sponsors and research sites, supervising the accuracy of data collected during studies.
What are the primary responsibilities of a CRA?
The primary responsibilities of a CRA include conducting site visits, reviewing patient records, and ensuring that studies are executed ethically and safely.
Why is the oversight of CRAs important in medical research?
The oversight of CRAs is crucial for maintaining the quality and reliability of medical research, which directly impacts patient outcomes and the approval of new therapies.
What is the projected growth of the clinical trial market?
The clinical trial market is projected to exceed $80 billion by 2025, indicating a rising demand for skilled Clinical Research Associates.
What challenges do research organizations face in the clinical research sector?
Research organizations face challenges such as regulatory hurdles, competition, and high turnover rates in the research sector, which range from 24-29%.
How do CRAs contribute to the development of innovative therapies?
CRAs are indispensable in the research landscape as they ensure compliance and uphold ethical standards, contributing to the successful development of innovative therapies.
How are CRAs involved in promoting diversity in clinical studies?
As the industry emphasizes diversity in clinical studies, CRAs play a vital role in promoting inclusive study experiences, ensuring that research reflects the diverse populations it aims to serve.
What career path do many Clinical Research Associates follow?
Many Clinical Research Associates begin their careers as Research Coordinators or Trial Assistants, providing a pathway for professional growth within the field.
What services do CRAs need to navigate for successful trial outcomes?
CRAs must navigate comprehensive services including feasibility studies, site selection, compliance reviews, trial setup, import permits, project management, and reporting to secure successful trial outcomes.