


The role of a project manager in clinical research is paramount for ensuring the successful execution of research studies. They oversee planning, execution, and compliance with regulatory standards. Effective project management not only enhances collaboration but also mitigates risks and addresses challenges such as recruitment delays. Ultimately, these efforts lead to improved study outcomes and uphold the integrity of medical research.
The intricate world of clinical research hinges on the effectiveness of project management, a role that ensures studies are executed with precision and integrity. By navigating the complexities of timelines, budgets, and stakeholder communication, a clinical research project manager plays a crucial role in advancing medical knowledge and improving patient care. However, with approximately 80% of studies facing delays due to recruitment challenges, one must ask: what essential skills and responsibilities define a successful project manager in this high-stakes field?
The project manager in clinical research is pivotal in the successful implementation of research studies, overseeing the planning, execution, and monitoring of projects. As a project manager in clinical research, this role demands skilled coordination of various components, including timelines, budgets, and resources, to ensure that assessments are conducted efficiently and effectively.
The project manager in clinical research serves as an essential intermediary among stakeholders—sponsors, regulatory bodies, and research sites—ensuring alignment and clear communication throughout the study. Their guidance is critical for a project manager in clinical research to manage the complexities of clinical research, as evidenced by case studies that demonstrate how effective management can significantly enhance the success rates of studies.
For instance, projects with dedicated managers often experience improved collaboration and adherence to timelines, which are crucial for advancing medical knowledge and enhancing patient care. Notably, 80% of studies face delays due to recruitment challenges, highlighting the importance of a project manager in clinical research in addressing such issues.
By managing risks and ensuring compliance with regulatory standards, including the import permit process and compliance assessments, the project manager in clinical research facilitates smoother research operations and bolsters the overall integrity and credibility of medical studies. This is particularly significant in the context of bioaccess's extensive management services for studies, which encompass feasibility assessments, site selection, compliance evaluations, setup, import permits, management, and reporting.
Furthermore, partnerships like that of bioaccess and Caribbean Health Group to establish Barranquilla as a leading location for medical trials in Latin America illustrate the proactive role of the project manager in clinical research in enhancing recruitment strategies and operational efficiency, ultimately resulting in superior trial outcomes.
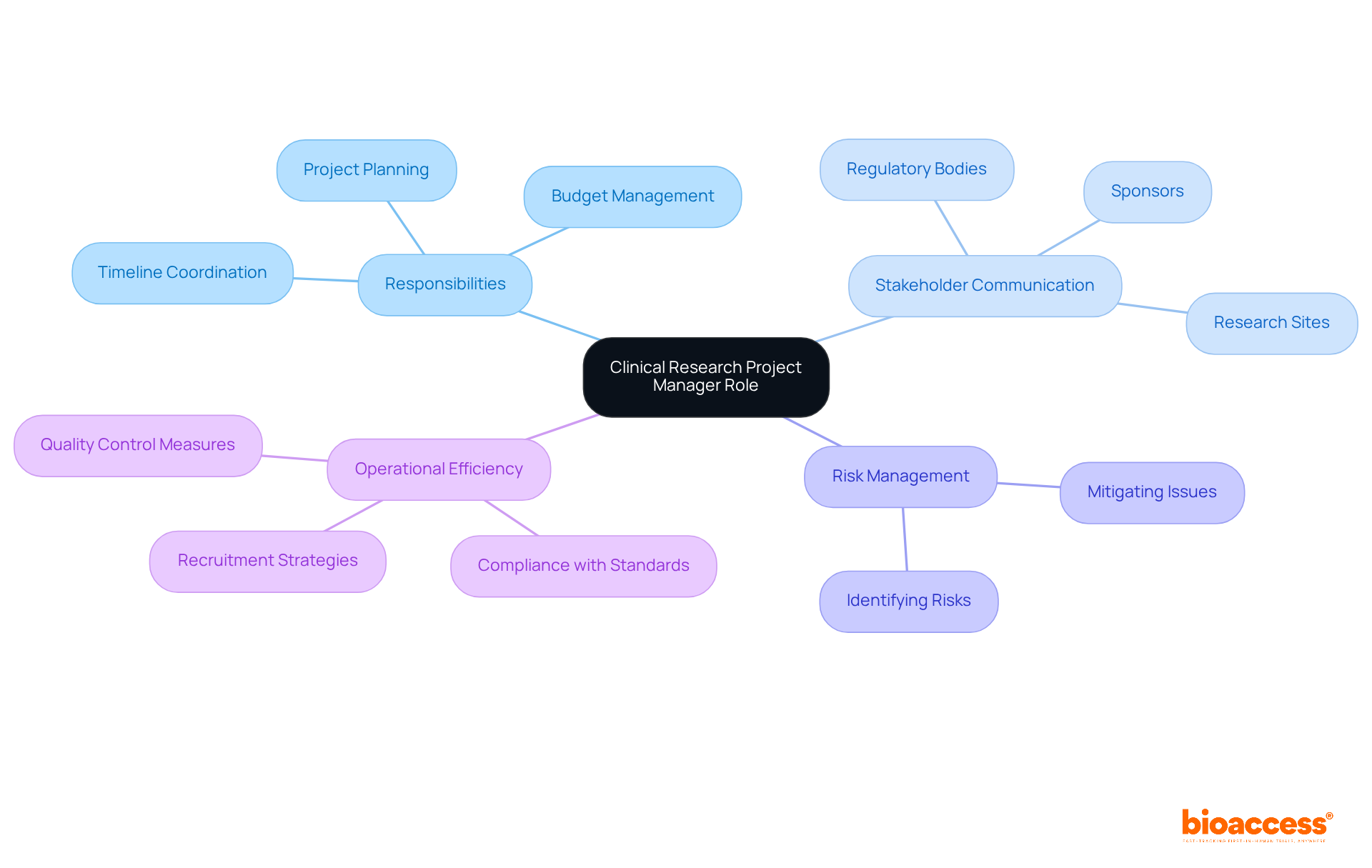
Key responsibilities of a project manager in clinical research encompass critical areas that are essential for effective research management.
Initiative Planning: Developing comprehensive plans that outline timelines, milestones, and resource distribution is essential. Efficient planning of initiatives is acknowledged as a key priority by 46% of organizations, highlighting its significance for a project manager in clinical research.
Team Leadership: As a project manager in clinical research, this role includes recruiting, training, and directing a team of researchers and medical personnel to foster effective collaboration. Robust guidance is crucial for a project manager in clinical research to manage the intricacies of medical studies, where 85% of managers frequently oversee multiple initiatives simultaneously.
Budget Monitoring: For a project manager in clinical research, monitoring budgets for initiatives is vital to ensure that financial resources are utilized efficiently. In 2025, the average funding allocation for research trials is anticipated to be considerable, making efficient budget management an essential skill for a project manager in clinical research.
Regulatory Compliance: A project manager in clinical research is responsible for ensuring adherence to regulatory guidelines and ethical standards, which is a fundamental duty. This compliance is essential for maintaining the integrity of clinical research and is a key responsibility of the project manager in clinical research to safeguard participant safety.
Stakeholder Communication: Maintaining clear and consistent communication with all stakeholders is necessary for a project manager in clinical research to keep them informed of progress and challenges. Effective communication can significantly decrease failures in endeavors, as miscommunication contributes to 19% of such failures.
Risk Management: As a project manager in clinical research, identifying potential risks and developing strategies to mitigate them is essential for achieving success. Organizations that implement robust risk management practices are better positioned to navigate the uncertainties inherent in clinical research.
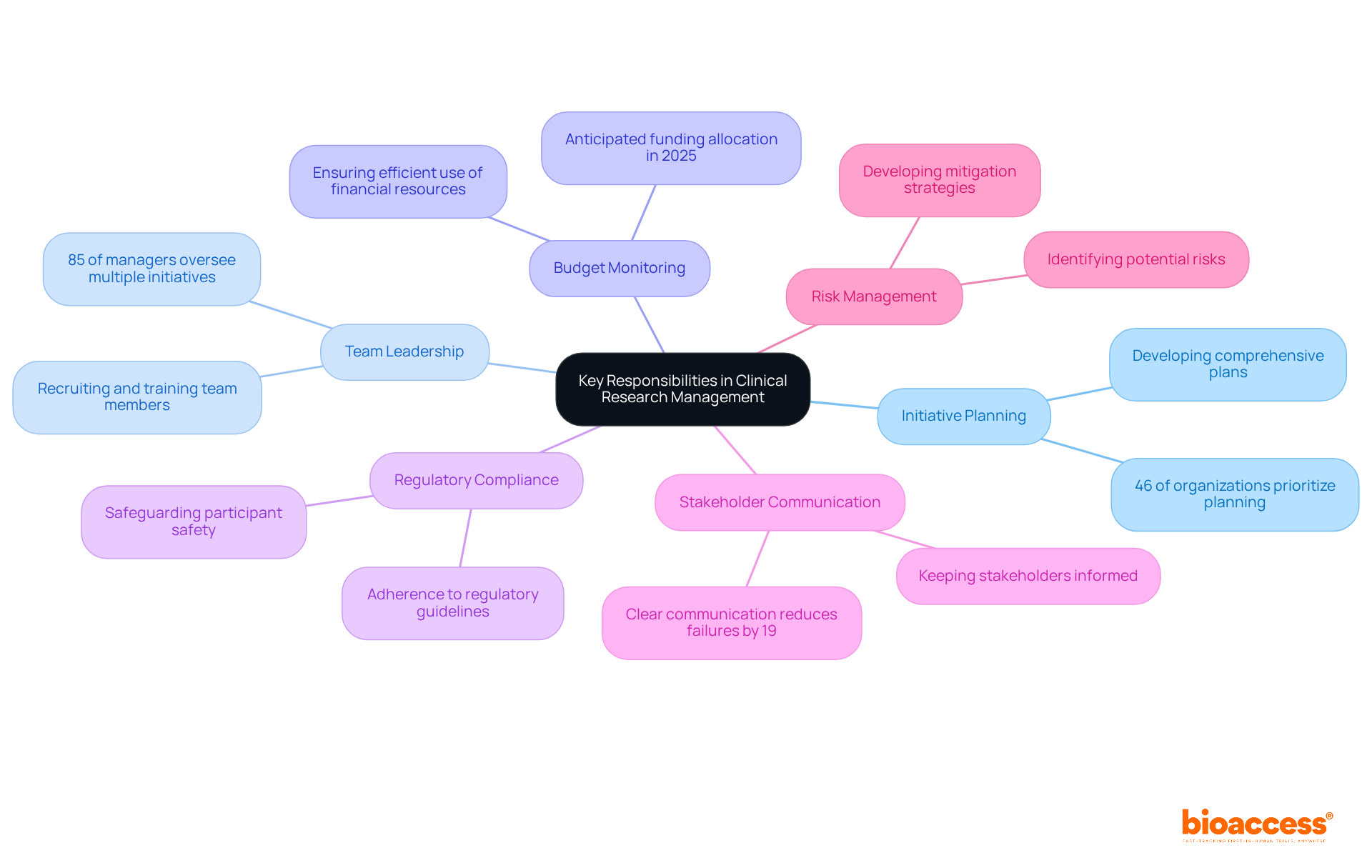
Essential skills for effective Clinical Research Project Management encompass several critical areas:
Leadership: The capacity to motivate and direct a team towards accomplishing objectives is essential. Effective leaders foster collaboration and inspire team members, ensuring alignment with the project's objectives.
Communication: Strong verbal and written communication skills are vital for facilitating collaboration among diverse stakeholders, including sponsors, investigators, and regulatory agencies. Clear communication enhances understanding and minimizes misunderstandings, which can significantly impact trial outcomes.
Analytical Thinking: The ability to analyze data and make informed decisions based on research findings is crucial. Managers must interpret intricate data sets to steer initiative direction and ensure adherence to regulatory standards. With bioaccess's accelerated regulatory approval process, which can take as little as 6-8 weeks compared to 6-12 months in the US/EU, project managers can navigate these complexities more efficiently.
Problem-Solving: The capability to recognize problems swiftly and create efficient solutions is vital in the ever-changing landscape of medical studies. Proactive problem-solving assists in reducing risks and preserving study integrity, especially in addressing patient recruitment difficulties in early-stage research.
Attention to Detail: Ensuring accuracy in documentation and adherence to protocols is critical for maintaining data integrity. A meticulous approach helps prevent errors that could jeopardize study results and participant safety. For instance, the role of Clinical Research Coordinators (CRCs) in guaranteeing patient safety underscores the significance of meticulousness in clinical studies.
Time Management: The ability to prioritize tasks and manage multiple initiatives concurrently is essential for ensuring the timely completion of all stages of the examination. Effective time management enables managers to meet deadlines and sustain momentum. With bioaccess's expertise in managing Early-Feasibility, First-In-Human, Pilot, Pivotal, and Post-Market Follow-Up Studies, project managers can leverage these skills to enhance study efficiency.
Regulatory compliance and ethical standards serve as the bedrock of effective clinical research management. A project manager in clinical research must ensure that all studies comply with both local and international regulations, including Good Clinical Practice (GCP) and the Declaration of Helsinki. This process necessitates obtaining required approvals from ethics committees (ECs) prior to commencing the study, alongside ensuring that ECs conduct initial and ongoing evaluations at appropriate intervals.
Ethical considerations are paramount, emphasizing the informed consent process, participant safety, and data confidentiality. For instance, the informed consent document must explicitly outline the study's purpose, procedures, risks, benefits, and confidentiality measures, including a statement regarding the availability of study information on ClinicalTrials.gov, thereby ensuring participants are fully informed and their rights are protected.
Statistics reveal that a mere three percent of U.S. doctors and patients engage in research studies, underscoring the critical need for robust ethical practices to cultivate trust and encourage participation. By rigorously adhering to these standards, the project manager in clinical research not only protects the rights and welfare of participants but also bolsters the credibility and reliability of research findings.
Furthermore, bioaccess offers comprehensive research management services, encompassing feasibility assessments, site selection, compliance evaluations, setup, import permits, project oversight, and reporting, all essential for ensuring regulatory adherence. The FDA's Bioresearch Monitoring (BIMO) Program plays a pivotal role in safeguarding human study participants and ensuring the reliability of medical study data.
Case studies, such as those examining the ethical principles guiding trials globally, underscore the significance of maintaining fairness and safety in investigative practices. Ultimately, a steadfast commitment to ethical standards enhances the overall integrity of clinical research, paving the way for significant advancements in medical science.

The role of a project manager in clinical research is indispensable, acting as the linchpin that connects various stakeholders and ensures the smooth and effective execution of research studies. This position demands a unique blend of leadership, communication, and analytical skills to navigate the complexities of clinical trials, ultimately driving advancements in medical science and enhancing patient care.
Throughout this article, we have highlighted key responsibilities of clinical research project managers, including:
Each of these elements plays a critical role in ensuring that clinical trials not only meet their objectives but also adhere to ethical standards and regulatory requirements. The significance of these responsibilities is further underscored by statistics indicating that effective project management can significantly mitigate common challenges, such as recruitment delays and miscommunication.
In conclusion, the importance of proficient project management in clinical research cannot be overstated. By fostering collaboration, ensuring compliance, and maintaining ethical integrity, project managers are essential in enhancing the credibility and reliability of medical studies. As the landscape of clinical research continues to evolve, prioritizing the development of these professionals will be crucial for driving innovation and improving health outcomes globally. Emphasizing the importance of strong project management practices will not only benefit individual studies but will also contribute to the broader mission of advancing medical knowledge and ensuring patient safety.
What is the role of a Clinical Research Project Manager?
The Clinical Research Project Manager is responsible for overseeing the planning, execution, and monitoring of research studies, ensuring that projects are conducted efficiently and effectively.
What are the key responsibilities of a Clinical Research Project Manager?
Key responsibilities include coordinating timelines, budgets, and resources, facilitating communication among stakeholders, managing risks, ensuring regulatory compliance, and addressing recruitment challenges.
How does a Clinical Research Project Manager contribute to study success?
A Clinical Research Project Manager enhances study success by improving collaboration, adherence to timelines, and effectively managing the complexities of clinical research, which can lead to higher success rates.
What challenges do Clinical Research Project Managers help address?
They help address challenges such as recruitment difficulties, compliance with regulatory standards, and risks associated with research operations.
Why is recruitment a significant concern in clinical studies?
Recruitment is a significant concern because 80% of studies face delays due to recruitment challenges, making effective management crucial for timely project completion.
How do partnerships, like that of bioaccess and Caribbean Health Group, impact clinical research?
Such partnerships help establish leading locations for medical trials, enhance recruitment strategies, and improve operational efficiency, leading to better trial outcomes.
What services are included in the management of clinical studies by organizations like bioaccess?
Services include feasibility assessments, site selection, compliance evaluations, setup, import permits, management, and reporting.