


The Trial Master File (TMF) serves as a vital repository in clinical trials, consolidating essential documents to uphold regulatory compliance and ensure data integrity throughout the study process. A well-maintained TMF is not merely beneficial; it is indispensable during audits and inspections. It provides a transparent record of all activities, which enhances the quality of clinical research and safeguards participant safety. This underscores the TMF's critical role in fostering trust and accountability within the clinical research landscape.
The Trial Master File (TMF) serves as a cornerstone in the realm of clinical trials, representing a comprehensive collection of essential documents that uphold compliance and integrity throughout the research process. As the complexity of clinical studies continues to expand, grasping the TMF's key components and its pivotal role becomes increasingly vital for stakeholders engaged in clinical research.
With the transition from traditional paper-based systems to digital formats, what challenges and opportunities emerge for maintaining an effective TMF that meets regulatory standards?
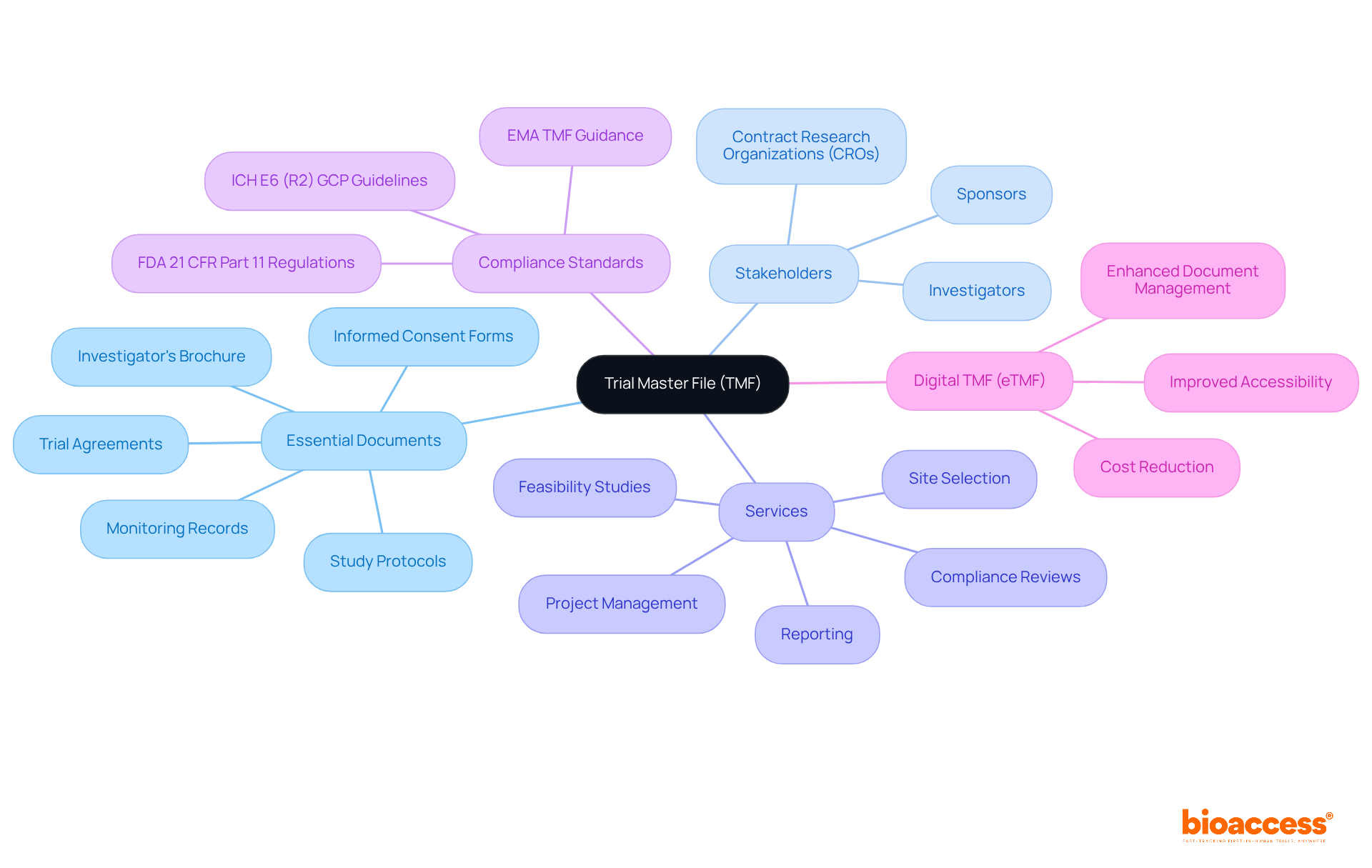
The TMF meaning refers to the Trial Master File, which serves as a crucial repository that consolidates all essential documents necessary to demonstrate compliance with standards throughout a clinical study. This comprehensive compilation includes vital documents such as study protocols, informed consent forms, approvals, and communications between stakeholders. Managed collaboratively by investigators, sponsors, and contract research organizations (CROs), the TMF meaning refers to the system that ensures all records are meticulously updated and readily accessible during the study process. Its significance is paramount, as the TMF meaning serves as an indispensable resource during oversight inspections and audits, providing a thorough account of the study's implementation and adherence to Good Clinical Practice (GCP).
Understanding the TMF meaning is essential for efficient management, as it enhances data integrity and supports compliance with evolving legal obligations, ensuring that all necessary documents are systematically included and organized. At bioaccess, our service capabilities encompass:
All of which are critical to maintaining an effective TMF. Regular evaluations of the TMF meaning are essential to ensure it remains inspection-ready, consistently conforming to compliance standards such as ICH E6 (R2) Good Clinical Practice (GCP) guidelines and FDA 21 CFR Part 11 regulations.
Understanding the retention period for clinical research data, which has been reduced to 15 years by Health Canada, is crucial for effective TMF management. The transition to a digital TMF (eTMF) presents advantages such as improved accessibility and cost reduction, reflecting contemporary practices in clinical research management. Katherine Ruiz, a specialist in oversight matters for medical apparatus and in vitro diagnostics in Colombia, underscores the importance of these procedures in ensuring adherence and effectiveness in clinical studies.
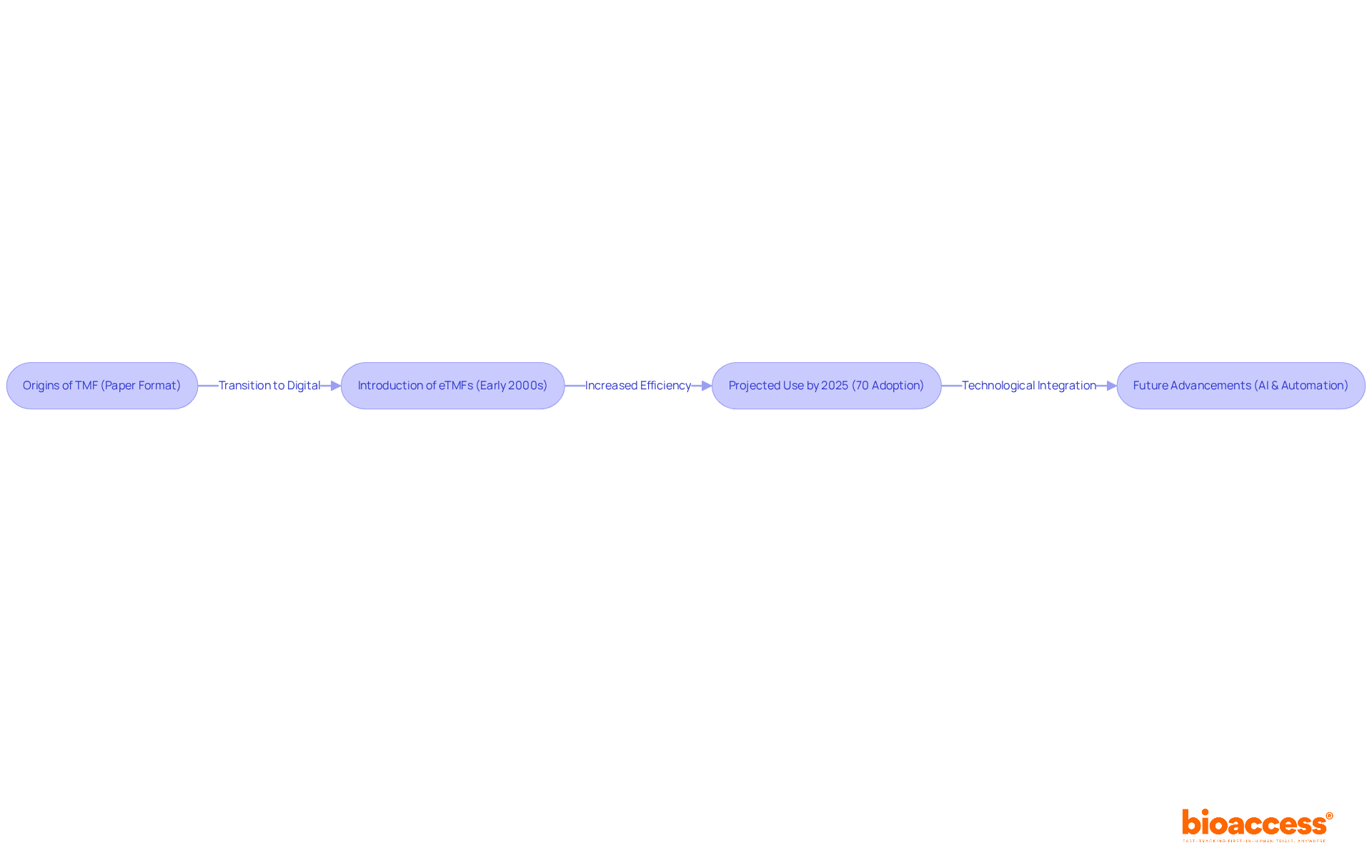
The concept of TMF meaning emerged as a solution to the increasing complexity of clinical studies and the urgent need for standardized documentation. Initially, TMFs were maintained in paper format, which resulted in disorganized and cumbersome files that hindered efficiency. Recognizing the critical role of a well-organized TMF meaning in ensuring adherence and data integrity, regulatory bodies began advocating for necessary enhancements.
The introduction of electronic Trial Master Files (eTMFs) in the early 2000s marked a pivotal shift, facilitating more efficient document management, real-time updates, and improved accessibility. By 2025, it is projected that over 70% of clinical studies will employ eTMFs, highlighting a significant trend toward digital solutions in clinical research.
The evolution of the TMF meaning continues with advancements in technology, such as the integration of artificial intelligence and automated workflows, which further enhance adherence and streamline process management. Organizations like bioaccess leverage these innovations in their comprehensive clinical study management services, which include setup and compliance evaluations, to ensure that TMFs are managed efficiently and in alignment with compliance standards.
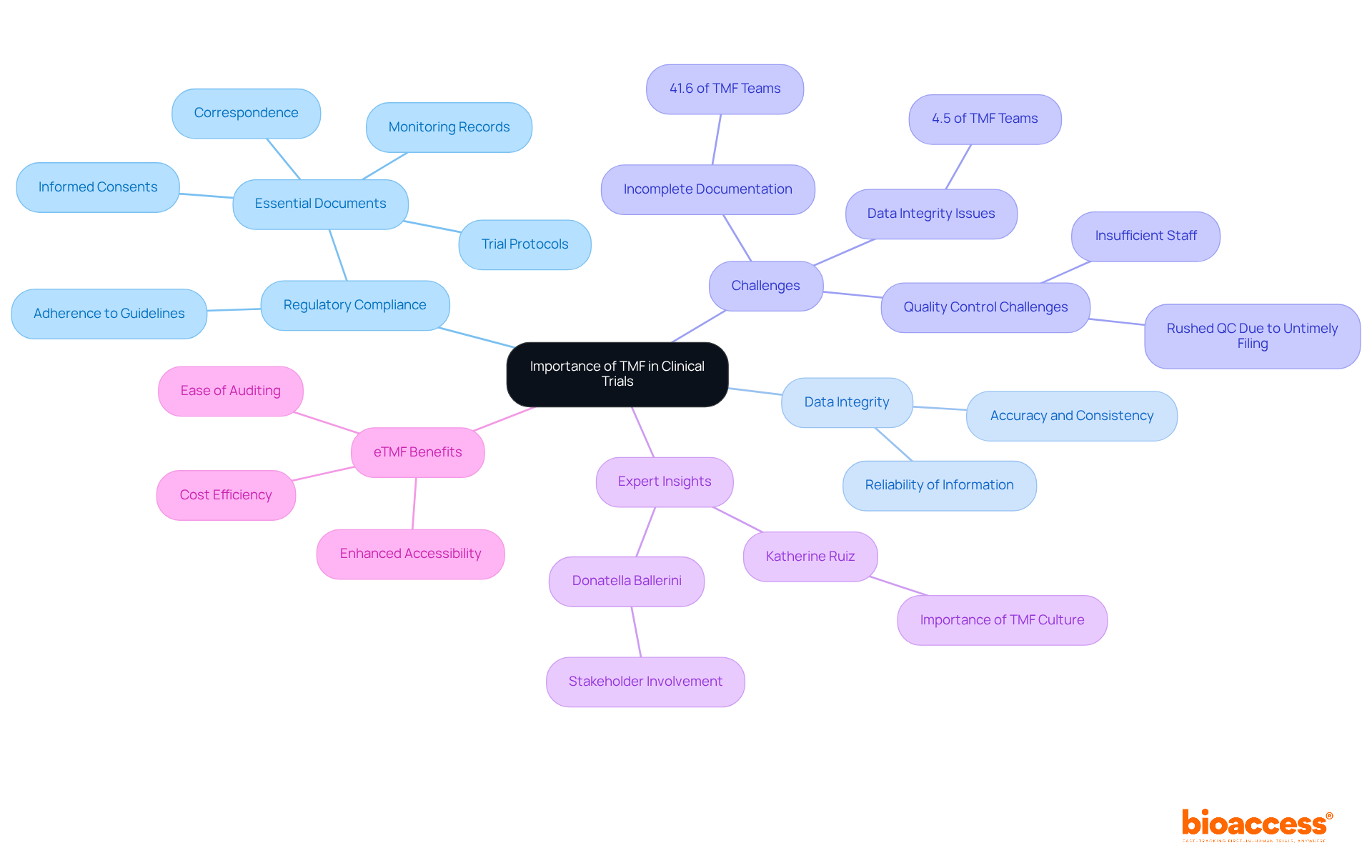
The Trial Master File (TMF) serves as a crucial repository in clinical research, housing essential documents such as study protocols, informed consent forms, approvals, and monitoring reports, which illustrates the TMF meaning. Additionally, it encompasses management documents like delegation logs, training records, and communications with oversight bodies. Each document is integral to upholding the integrity and transparency of the research process.
For efficient audits and inspections, the TMF must be meticulously organized, typically categorized by document type and maintained in chronological order. This structured approach not only facilitates straightforward access and review but also enhances compliance with legal requirements. Best practices for organizing TMF documents involve:
Expert opinions underscore that a well-maintained TMF can significantly elevate data quality and integrity, ultimately contributing to the successful execution of clinical studies. Typically, the TMF meaning for clinical studies comprises numerous vital documents, underscoring the complexity and compliance demands of the research process. Regular audits and updates are indispensable for preserving the TMF's accuracy and completeness, thus reinforcing its role as a cornerstone of legal adherence.

The Trial Master File (TMF) is essential for understanding TMF meaning, as it plays a pivotal role in maintaining regulatory compliance and safeguarding the integrity of clinical research data. It functions as the primary repository for documentation, providing a transparent record of all activities that adhere to established guidelines and standards. A meticulously maintained TMF meaning significantly enhances data integrity by ensuring accurate documentation of all processes, which is vital for evaluating study outcomes.
Furthermore, the TMF is indispensable during audits and inspections by oversight bodies, offering a comprehensive account of the study's execution. Insufficient oversight of the TMF can result in compliance issues, data inconsistencies, and potential delays in approval processes. Statistics reveal that:
Therefore, understanding the TMF meaning is crucial for upholding a high-quality TMF, which is essential not only for the successful execution of clinical studies but also for advancing medical knowledge and ensuring participant safety.
Expert insights underscore that cultivating a robust TMF culture, supported by experienced professionals like Katherine Ruiz, a specialist in regulatory affairs for medical devices and in vitro diagnostics in Colombia, is essential for preempting and addressing potential issues, thereby enhancing overall study outcomes. As emphasized by Donatella Ballerini, the involvement of all relevant TMF stakeholders is vital for completeness and adherence.
Additionally, transitioning to electronic TMF (eTMF) systems presents advantages such as enhanced accessibility and cost efficiency, further bolstering effective TMF management. Comprehensive clinical study management services—including feasibility assessments, site selection, regulatory reviews, study setup, import permits, project management, and reporting—are critical in ensuring the TMF's integrity and compliance.
Case studies further illustrate the TMF's essential role in ensuring compliance and data integrity, reinforcing its significance in the clinical trial landscape.
The Trial Master File (TMF) stands as a foundational element in clinical research, serving as a comprehensive repository of essential documents that ensure compliance with regulatory requirements and uphold the integrity of study data. Understanding the TMF's significance is vital for all stakeholders involved in clinical trials, as it encapsulates the collaborative efforts necessary for successful study execution and adherence to Good Clinical Practice (GCP).
Key insights from the article underscore the TMF's pivotal role in maintaining data integrity, the transition from traditional paper formats to electronic systems, and the critical importance of meticulous organization and regular updates. The TMF not only supports compliance during audits and inspections but also enhances the overall quality of clinical studies, thereby contributing to the advancement of medical knowledge and participant safety.
Given the complexities surrounding clinical trials, it is imperative for organizations to prioritize effective TMF management. Embracing digital solutions and fostering a culture of thorough documentation will significantly mitigate compliance risks and streamline processes. By understanding and implementing best practices related to the TMF, stakeholders can ensure that clinical research not only meets regulatory standards but also achieves its ultimate goal of improving health outcomes.
What is the Trial Master File (TMF)?
The Trial Master File (TMF) is a crucial repository that consolidates all essential documents necessary to demonstrate compliance with standards throughout a clinical study. It includes vital documents such as study protocols, informed consent forms, approvals, and communications between stakeholders.
Who manages the TMF?
The TMF is managed collaboratively by investigators, sponsors, and contract research organizations (CROs) to ensure that all records are meticulously updated and readily accessible during the study process.
Why is the TMF important?
The TMF is important because it serves as an indispensable resource during oversight inspections and audits, providing a thorough account of the study's implementation and adherence to Good Clinical Practice (GCP).
How does the TMF enhance data integrity?
Understanding the TMF is essential for efficient management, as it enhances data integrity and supports compliance with evolving legal obligations, ensuring that all necessary documents are systematically included and organized.
What services are included in maintaining an effective TMF?
Services critical to maintaining an effective TMF include feasibility studies, site selection, compliance reviews, setup, import permits, project management, and reporting.
What compliance standards must the TMF conform to?
The TMF must consistently conform to compliance standards such as ICH E6 (R2) Good Clinical Practice (GCP) guidelines and FDA 21 CFR Part 11 regulations.
What is the retention period for clinical research data according to Health Canada?
The retention period for clinical research data has been reduced to 15 years by Health Canada, which is crucial for effective TMF management.
What are the advantages of transitioning to a digital TMF (eTMF)?
The transition to a digital TMF (eTMF) presents advantages such as improved accessibility and cost reduction, reflecting contemporary practices in clinical research management.