


Excipients are inactive substances in pharmaceuticals that play crucial roles in enhancing the efficacy, stability, and safety of medications, despite lacking therapeutic effects themselves. Their importance cannot be overstated; excipients improve drug bioavailability, facilitate manufacturing processes, and enhance patient compliance. However, the careful selection of these substances is essential to avoid potential adverse reactions. This highlights the need for a thorough understanding of their functions and implications in clinical research.
In the intricate world of pharmaceuticals, excipients often remain in the shadows; yet, their influence is profound. These inactive ingredients, while devoid of therapeutic properties, are essential for enhancing the stability, bioavailability, and overall effectiveness of medications.
As the pharmaceutical landscape evolves, understanding the multifaceted roles of excipients becomes crucial, particularly given their potential impact on patient safety and treatment outcomes.
What challenges arise when these seemingly benign substances provoke adverse reactions? How can their selection be optimized to ensure both efficacy and safety?
Delving into the realm of excipients reveals a complex interplay of science, regulation, and patient care, inviting exploration into their critical functions in drug formulation.
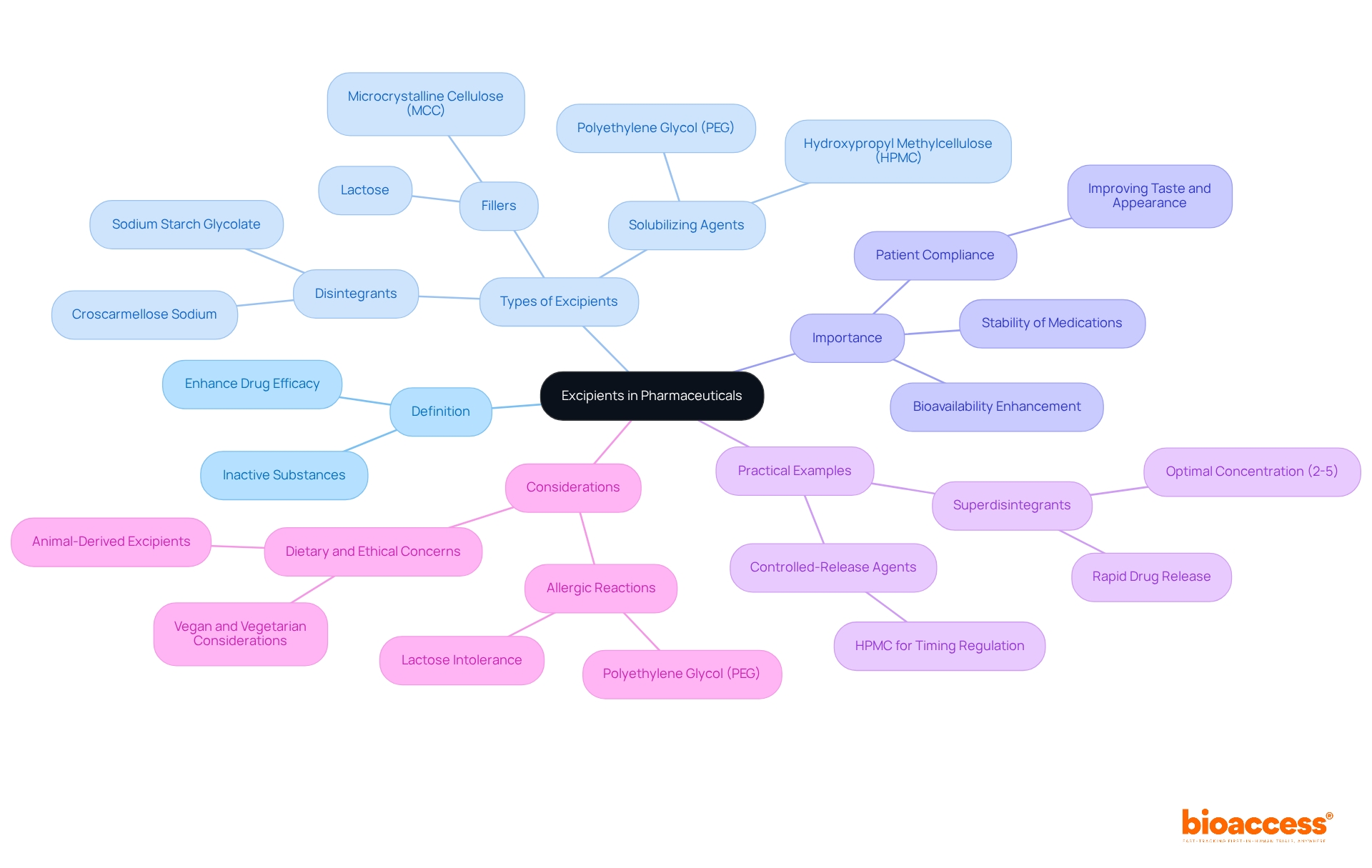
What is excipient in medicinal formulations refers to integral substances that, while lacking therapeutic effects, perform essential functions enhancing the overall efficacy and safety of medications. They play a pivotal role in the manufacturing process, contributing to the stability and bioavailability of active pharmaceutical ingredients (APIs). For instance, substances like polyethylene glycol (PEG) serve as solubilizing agents to enhance medication dissolution, which is crucial for ensuring that treatments are effective upon administration.
The variety of additives encompasses:
Statistics indicate that additives significantly influence the success of medications; they can enhance the bioavailability of poorly water-soluble drugs, with estimates suggesting that over 40% of drugs on the market and nearly 90% of molecules in the discovery pipeline are poorly soluble. This underscores what is excipient and its essential function in enhancing the effectiveness of these medications.
As we move towards 2025, understanding what is excipient becomes increasingly important, as these inactive ingredients are recognized not merely as fillers but as essential elements that guarantee the stability and effectiveness of pharmaceutical products. Their roles extend to improving patient compliance by enhancing the taste and appearance of medications, thereby promoting adherence to treatment regimens. Experts in the field emphasize that understanding what is excipient is vital for the careful selection and application of additives to create therapies that are effective and meet regulatory safety standards.
Practical instances illustrate the influence of additives on medication effectiveness. For example, the application of superdisintegrants at optimal concentrations (usually between 2-5% of the tablet weight) can result in over 90% of the active ingredient being released within 15 minutes, significantly enhancing therapeutic outcomes. Additionally, substances such as Hydroxypropyl Methylcellulose (HPMC) are utilized not only as controlled-release agents but also as binders, regulating the timing of release and ensuring stable plasma concentrations.
In summary, additives are crucial in contemporary medication formulation and delivery, enhancing stability, bioavailability, and patient experience while ensuring compliance with health regulations. However, it is important to recognize that some additives may provoke adverse reactions in sensitive individuals, necessitating rigorous quality control measures to ensure their safety. Furthermore, considerations regarding dietary, ethical, or religious beliefs may influence the selection of excipients in pharmaceutical formulations.
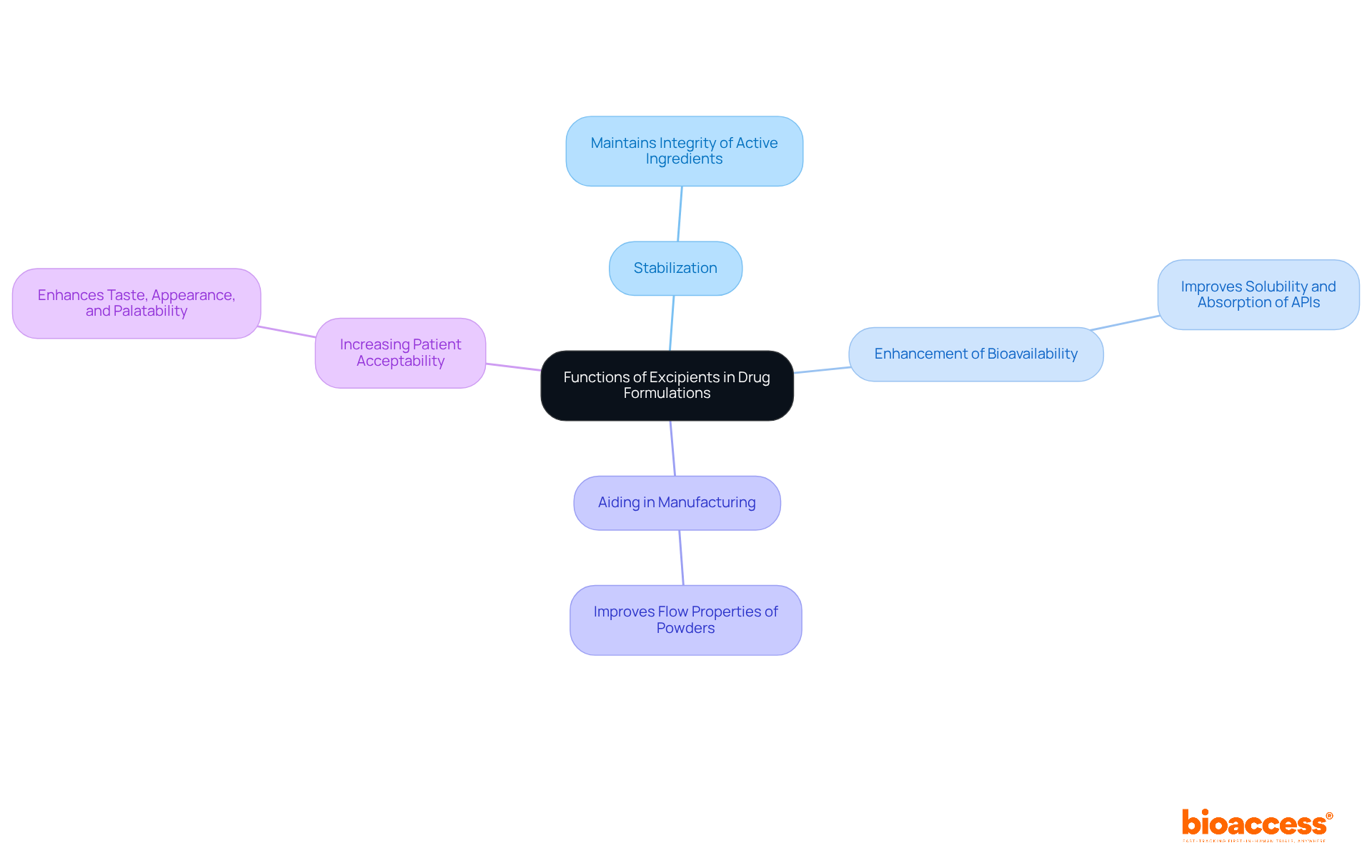
Understanding what is excipient is crucial as excipients serve multiple functions in drug formulations, including:
These functions emphasize what is excipient, illustrating the essential role that inactive ingredients play in ensuring that pharmaceuticals are not only effective but also safe and user-friendly.
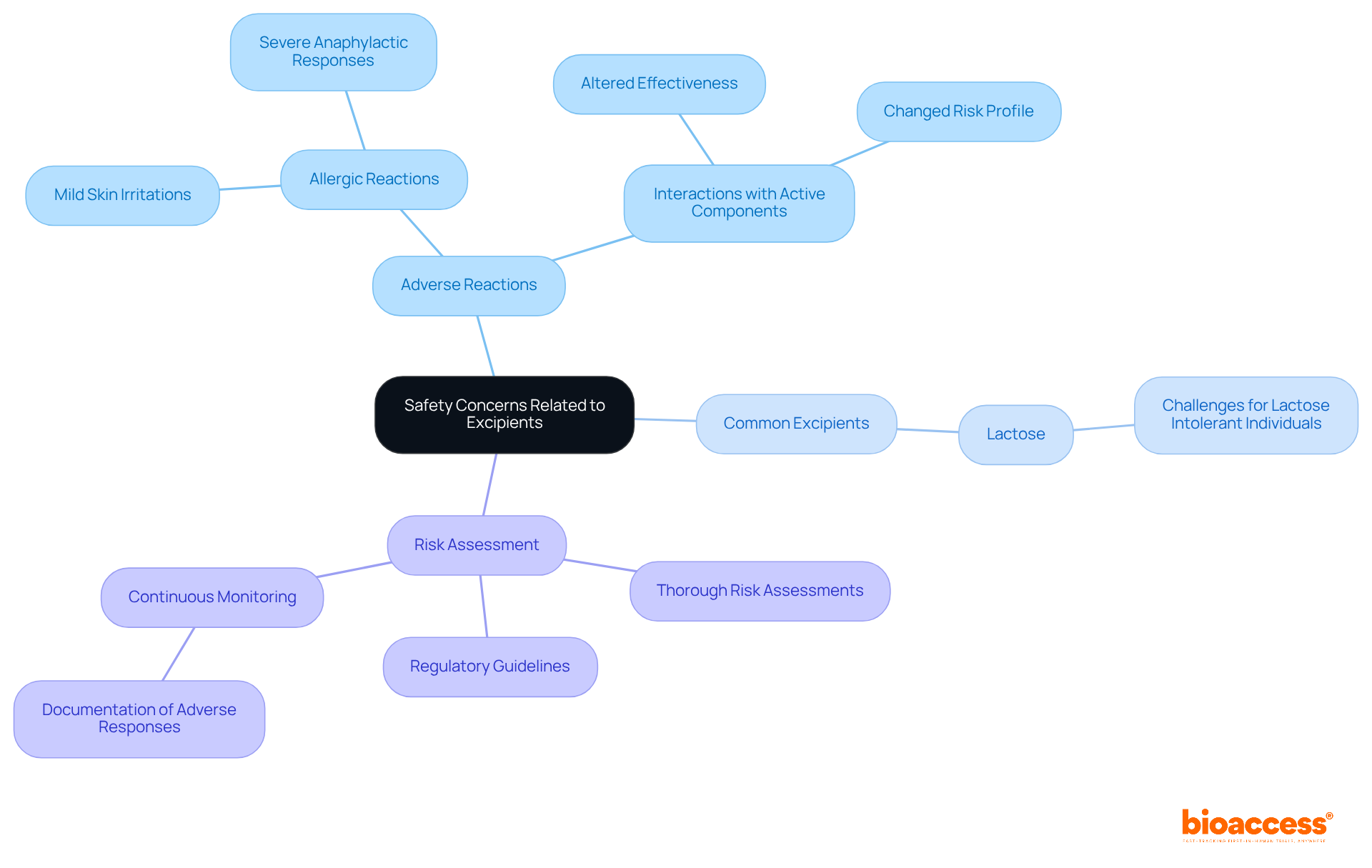
While inactive ingredients are generally regarded as safe, they can, on occasion, lead to negative reactions in certain individuals. Common issues encompass allergic reactions, which may vary from mild skin irritations to severe anaphylactic responses.
For instance, lactose, a prevalent excipient, highlights the importance of understanding what is excipient, as it poses challenges for those with lactose intolerance. Moreover, certain additives can interact with active components, potentially altering their effectiveness or risk profile.
Therefore, it is imperative for pharmaceutical producers to conduct thorough risk assessments and adhere to regulatory guidelines to mitigate these hazards. Continuous monitoring and documentation of adverse responses related to additives are also essential to ensure patient well-being.
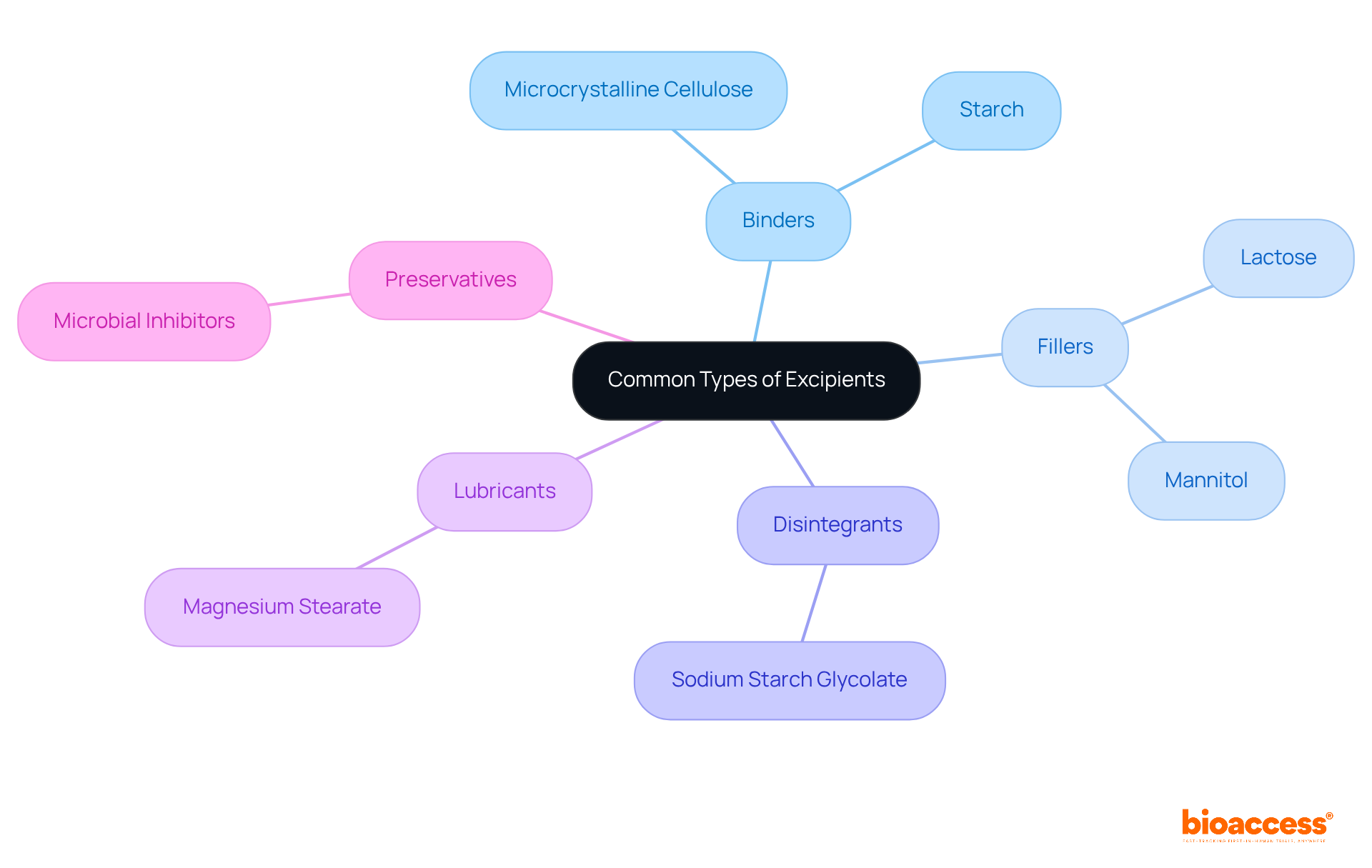
Common types of excipients include:
Understanding these categories is essential for appreciating how excipients contribute to the overall effectiveness and safety of pharmaceutical products.
Excipients are far more than mere fillers in pharmaceutical formulations; they are essential components that significantly enhance the effectiveness, stability, and safety of medications. Their multifaceted roles—from improving bioavailability and patient compliance to ensuring the integrity of active ingredients—underscore the importance of understanding what excipients are and how they function within drug products.
Throughout the article, various types of excipients have been explored, including:
Each category plays a crucial role in the formulation process, contributing to the overall therapeutic outcome and patient experience. Additionally, the discussion on safety highlights the necessity for rigorous quality control and risk assessment, as some excipients can provoke adverse reactions in sensitive individuals.
Recognizing the significance of excipients in pharmaceuticals is vital for both manufacturers and consumers alike. As the industry advances and the complexity of drug formulations increases, a deeper understanding of these inactive ingredients will be essential to ensure the development of safe, effective, and patient-friendly medications. Emphasizing the need for careful selection and regulation of excipients can lead to improved health outcomes and foster greater confidence in pharmaceutical products.
What are excipients in pharmaceuticals?
Excipients are integral substances in medicinal formulations that, while lacking therapeutic effects, perform essential functions to enhance the overall efficacy and safety of medications. They contribute to the stability and bioavailability of active pharmaceutical ingredients (APIs).
What roles do excipients play in medication formulations?
Excipients enhance medication dissolution, improve stability, increase bioavailability, and can improve patient compliance by enhancing the taste and appearance of medications.
Can you provide examples of common excipients?
Common excipients include fillers like lactose, disintegrants such as croscarmellose sodium, and solubilizing agents like polyethylene glycol (PEG).
Why are excipients important for poorly water-soluble drugs?
Excipients significantly influence the success of medications, especially for poorly water-soluble drugs. Over 40% of drugs on the market and nearly 90% of molecules in the discovery pipeline are poorly soluble, and excipients can enhance their bioavailability.
How do excipients affect patient compliance?
Excipients can improve the taste and appearance of medications, which promotes adherence to treatment regimens and enhances the overall patient experience.
What is the significance of understanding excipients in pharmaceutical development?
Understanding excipients is vital for the careful selection and application of additives to create effective therapies that meet regulatory safety standards and ensure the stability and effectiveness of pharmaceutical products.
What are some practical examples of excipient application in medications?
The use of superdisintegrants at optimal concentrations can result in over 90% of the active ingredient being released within 15 minutes. Hydroxypropyl Methylcellulose (HPMC) is used as both a controlled-release agent and a binder to regulate drug release timing.
Are there any safety concerns associated with excipients?
Yes, some excipients may provoke adverse reactions in sensitive individuals, highlighting the need for rigorous quality control measures to ensure their safety.
How can dietary, ethical, or religious beliefs influence excipient selection?
Considerations regarding dietary, ethical, or religious beliefs may impact the choice of excipients in pharmaceutical formulations, necessitating careful selection to accommodate these factors.