

Medical device classification is crucial for regulatory success, as it categorizes devices based on risk and dictates the compliance obligations that manufacturers must fulfill before marketing. Understanding this classification framework is not merely a regulatory requirement; it is vital for ensuring patient safety and significantly influences the efficiency and costs associated with bringing medical devices to market. Higher-risk categories necessitate more rigorous testing and oversight, underscoring the importance of compliance in the Medtech landscape. By grasping the intricacies of device classification, stakeholders can navigate the complexities of regulatory processes more effectively.
Medical device classification serves as a crucial pillar in the regulatory landscape, determining the categorization of medical products based on intended use and associated risks. This classification not only influences compliance obligations but also plays a pivotal role in shaping the market trajectory for manufacturers. As regulatory frameworks become increasingly complex, companies must effectively navigate these systems to ensure both safety and market success. Understanding the nuances of medical device classification is essential for overcoming the challenges posed by differing regulations, particularly between the FDA and EU, and for leveraging clinical research to enhance product approval pathways.
Medical device classification serves as a systematic method for categorizing medical equipment based on their intended use and associated risk levels. This categorization encompasses four distinct categories:
Understanding the medical device classification framework is crucial, as it dictates the compliance obligations that producers must fulfill before marketing their products. For example, Category I items typically face minimal oversight, requiring a Medical Device Establishment License (MDEL) for importation or sale, whereas Category III items undergo rigorous testing and approval processes, which can take an average of 3 to 7 years to navigate before reaching the market.
The ramifications of medical device classification extend far beyond mere regulatory compliance; they significantly impact market success. Devices classified as Class II or III often necessitate more extensive premarket submissions, influencing both timelines and costs. Recent regulatory updates have underscored the necessity for enhanced monitoring and reporting, particularly for higher-risk equipment, to ensure ongoing safety and effectiveness. As noted by R K Goyal, "Considering the increasing utilization of medical devices, strict standards are necessary to ensure that the devices are safe, thoroughly examined, and have minimal adverse reactions."
Experts assert that a well-defined medical device classification system not only safeguards patient safety but also facilitates a more efficient route to market. By aligning development strategies with categorization needs, manufacturers can adeptly navigate the compliance landscape, ultimately boosting their chances of success in a competitive environment. Therefore, medical equipment categorization transcends being a mere formal requirement; it stands as a fundamental element that shapes the trajectory of medical advancements.

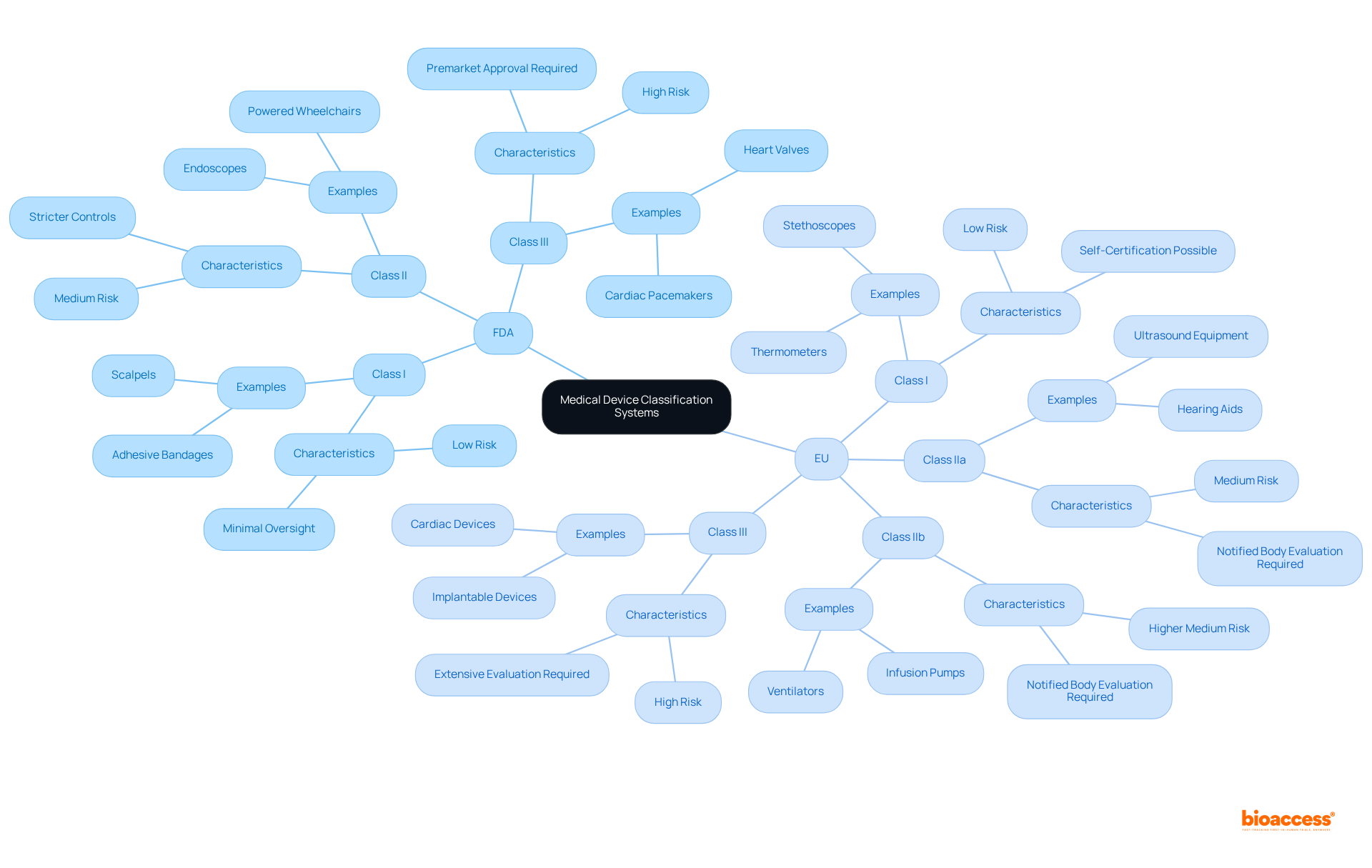
The medical device classification systems of the FDA and EU, both designed to ensure patient safety, reveal significant differences in their methodologies. The FDA employs medical device classification to categorize instruments into three classes: I, II, and III, based on the extent of regulatory oversight required to ensure safety and effectiveness.
Conversely, the medical device classification under the EU framework classifies instruments into four categories: I, IIa, IIb, and III, with Class I indicating the lowest risk and Class III the highest. The EU places considerable emphasis on conformity assessment procedures, which vary according to medical device classification.
Recent adjustments in FDA guidelines have aimed to streamline the 510(k) process, enhancing the predictability and efficiency of product approvals. Notably, 85 percent of FDA 510(k) applications received a Substantially Equivalent decision in 2022. However, it is crucial to note that 32% of submissions did not meet the minimum acceptability check, underscoring the challenges manufacturers face.
Producers seeking to penetrate both markets must navigate these compliance environments with caution, as the differences in medical device classification and evaluation processes significantly impact their oversight strategies and schedules. As Katelynn Larson, a technical writer, notes, "Manufacturers ought to assess the use-related risk and adjust their human factors engineering efforts according to the product’s classification and complexity." This perspective underscores the importance of aligning oversight methods with product characteristics to facilitate successful market entry. Furthermore, nearly 90% of medical equipment industry leaders now prioritize US regulatory approval over the EU, reflecting current industry trends.
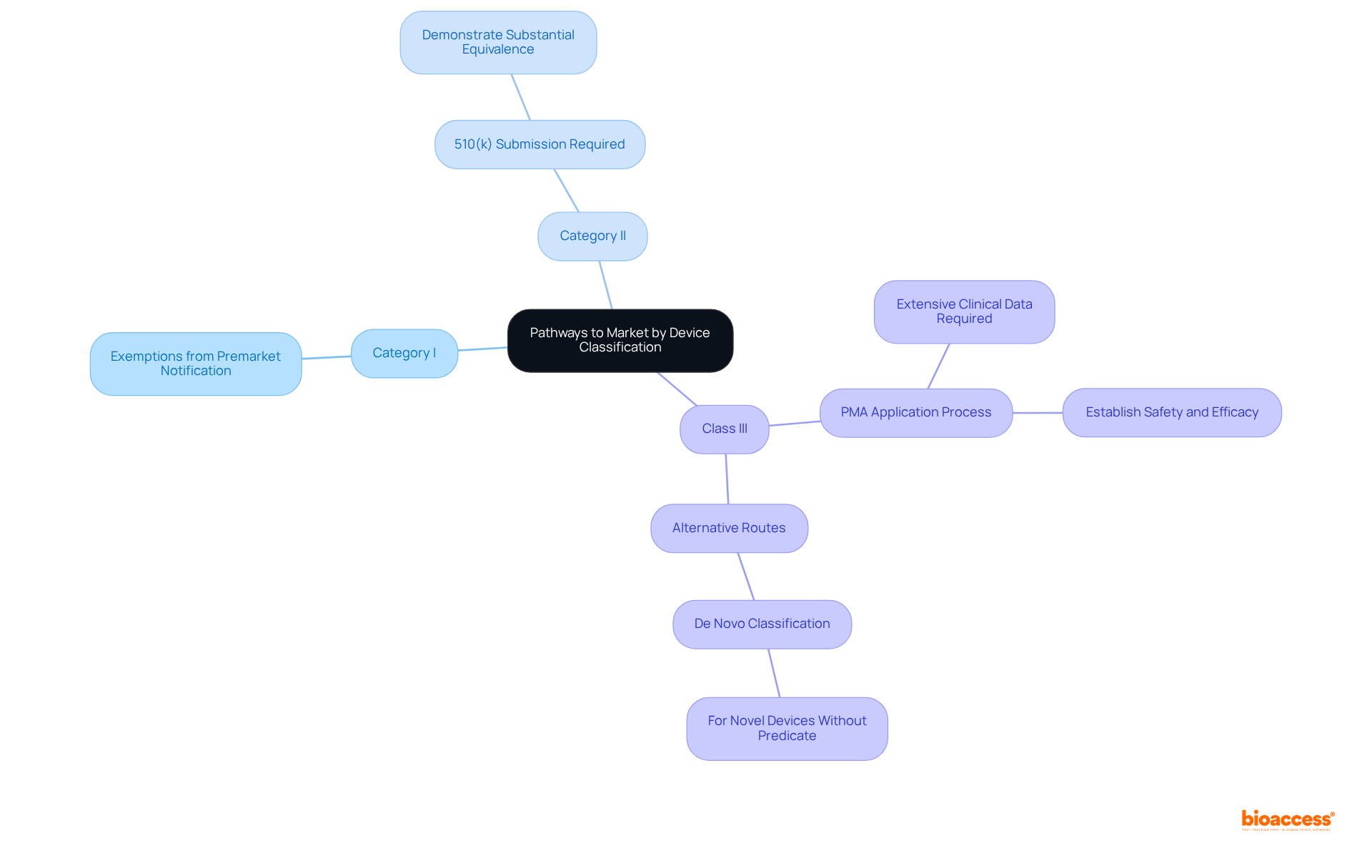
The route to market for medical products varies significantly based on their medical device classification. For Category I products, manufacturers often benefit from exemptions from premarket notification (510(k)), facilitating a more straightforward entry into the market. In contrast, Category II products typically require a 510(k) submission, demonstrating substantial equivalence to a legally marketed item.
For Class III products, the pathway becomes more intricate, necessitating a Premarket Approval (PMA) application. This process demands extensive clinical data to establish both safety and efficacy. Additionally, manufacturers may explore alternative routes, such as the De Novo classification for novel devices that do not have a predicate.
Understanding these regulatory pathways is crucial for manufacturers working within the framework of medical device classification. It enables them to effectively align their development timelines and allocate resources, ensuring compliance with regulatory requirements while minimizing time to market.
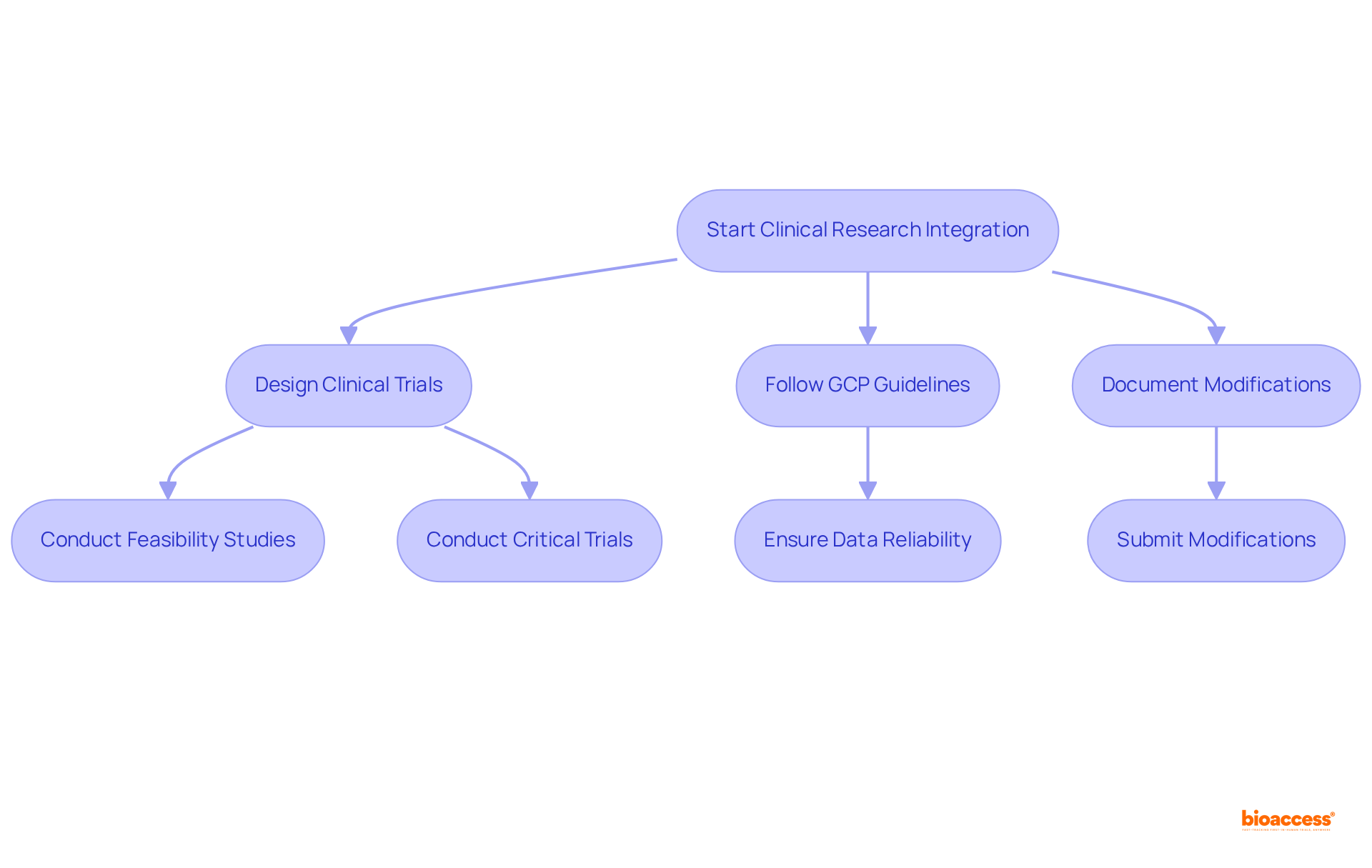
Clinical research is pivotal in the medical device classification process, particularly for higher-risk items classified as Class II and III. These instruments typically require robust clinical studies to gather evidence of safety and efficacy. Trials must be meticulously designed to comply with established standards, producing reliable data that supports the device's intended use.
Incorporating clinical research early in the product development cycle can significantly streamline the medical device classification process. By conducting feasibility studies and critical trials, manufacturers can proactively identify and address potential issues, thereby enhancing their chances of obtaining approval. Following Good Clinical Practice (GCP) guidelines, as outlined in ISO 14155:2020, is crucial, as it ensures the reliability of the data produced, which can withstand regulatory scrutiny.
Recent requirements for clinical trials have evolved, underscoring the necessity for comprehensive documentation and robust evidence. The Medical Device Regulation (MDR) stipulates that substantial modifications affecting safety or data validity must be documented and submitted. Additionally, the evaluation period for high-risk medical equipment can span 45 to 65 days, while verification for low-risk items may take 10 to 25 days. This regulatory environment emphasizes the importance of well-organized clinical trials in influencing medical equipment approval rates.
Manufacturers often face challenges in recruiting sufficient trial participants, particularly for high-risk products. Integrating clinical research not only aids in meeting compliance standards but also enhances the overall market success of medical products. By aligning clinical trials with regulatory expectations, manufacturers can more effectively navigate the complexities of medical device classification and approval.
Understanding the intricacies of medical device classification is essential for manufacturers aiming for regulatory success. This framework categorizes devices based on risk and dictates the compliance requirements necessary for market entry. The classification system significantly influences development timelines, costs, and overall market strategy, making it a fundamental aspect of the medical device industry.
The article highlights the critical distinctions between the FDA and EU classification systems, emphasizing how these differences impact regulatory oversight and market access. It delves into the pathways to market based on device classification, illustrating the varied requirements for low-risk versus high-risk products. Additionally, the role of clinical research is underscored as a pivotal factor in ensuring safety and efficacy, particularly for higher-risk devices that demand extensive clinical data.
In light of the evolving regulatory landscape, manufacturers are encouraged to adopt a proactive approach by aligning their development strategies with classification requirements. By integrating thorough clinical research and understanding the nuances of both FDA and EU regulations, organizations can enhance their chances of successful market entry. Ultimately, mastering medical device classification transcends mere compliance; it is a strategic imperative that drives innovation and improves patient safety in the ever-growing field of medical technology.
What is medical device classification?
Medical device classification is a systematic method for categorizing medical equipment based on their intended use and associated risk levels, which includes four categories: Category I (low risk), Category II (moderate risk), Category III (high risk), and Category IV (highest risk).
Why is medical device classification important?
Medical device classification is important because it dictates the compliance obligations that producers must fulfill before marketing their products. It also significantly impacts market success, influencing timelines and costs associated with bringing devices to market.
What are the different categories of medical devices?
The different categories of medical devices are: - Category I: Low risk - Category II: Moderate risk - Category III: High risk - Category IV: Highest risk
What are the compliance requirements for Category I devices?
Category I devices typically face minimal oversight, requiring a Medical Device Establishment License (MDEL) for importation or sale.
How long does it take for Category III devices to reach the market?
Category III devices undergo rigorous testing and approval processes, which can take an average of 3 to 7 years to navigate before reaching the market.
What recent updates have been made regarding medical device regulation?
Recent regulatory updates have emphasized the necessity for enhanced monitoring and reporting, particularly for higher-risk equipment, to ensure ongoing safety and effectiveness.
How does medical device classification affect manufacturers?
A well-defined medical device classification system helps manufacturers align their development strategies with categorization needs, aiding them in navigating the compliance landscape and increasing their chances of success in a competitive environment.
What is the broader impact of medical device classification on patient safety?
Medical device classification safeguards patient safety by ensuring that devices are safe, thoroughly examined, and have minimal adverse reactions, thus facilitating a more efficient route to market.