Overview
The article emphasizes the significance of essential clinical research management courses for directors aiming to elevate their leadership and operational capabilities within the field. It delineates various programs that concentrate on critical areas such as:
- Regulatory compliance
- Project management
- Quality assurance
- Recruitment strategies
These components collectively furnish leaders with the indispensable tools required to enhance research outcomes and adeptly navigate the complexities inherent in clinical trials.
Introducción
The landscape of clinical research is evolving rapidly, presenting both challenges and opportunities for directors leading these initiatives. As the demand for skilled leadership in this field grows, so too does the necessity for specialized training that equips professionals with the tools to navigate complex regulatory environments, enhance project management skills, and improve study outcomes.
What essential courses can directors pursue to not only stay ahead of industry trends but also ensure their teams thrive in this competitive arena?
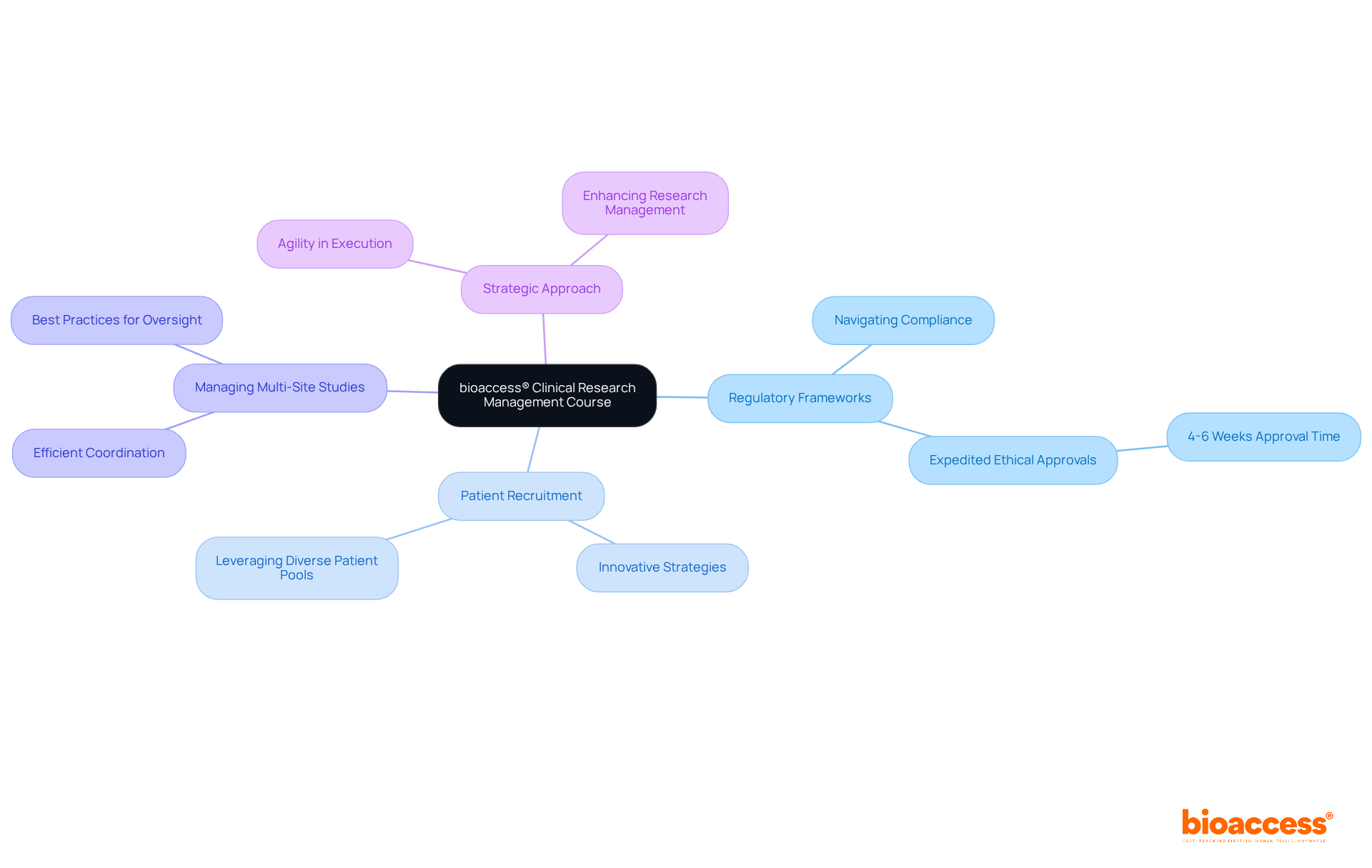
bioaccess® Clinical Research Management Course: Accelerate Your Career with Global-First Agility
The bioaccess® clinical research management course is specifically designed to tackle the unique challenges of conducting research studies across Latin America, Australia, and the Balkans. Participants will gain valuable insights into the regulatory efficiencies that bioaccess® leverages, achieving ethical approvals in just 4-6 weeks—significantly faster than traditional markets. This program is particularly tailored for leaders aiming to enhance their strategic approach to research management, emphasizing agility and efficiency in execution.
Key topics covered include:
- Navigating regional regulatory frameworks to ensure compliance and expedite processes.
- Innovative strategies for rapid patient recruitment, leveraging diverse patient pools.
- Best practices for managing multi-site studies efficiently, ensuring seamless coordination and oversight.
By completing the clinical research management course, directors will be empowered to guide their teams with a mindset centered on global-first agility, ultimately accelerating and enhancing research studies.
Certified Clinical Research Project Manager (CCRP™): Master Project Management in Clinical Trials
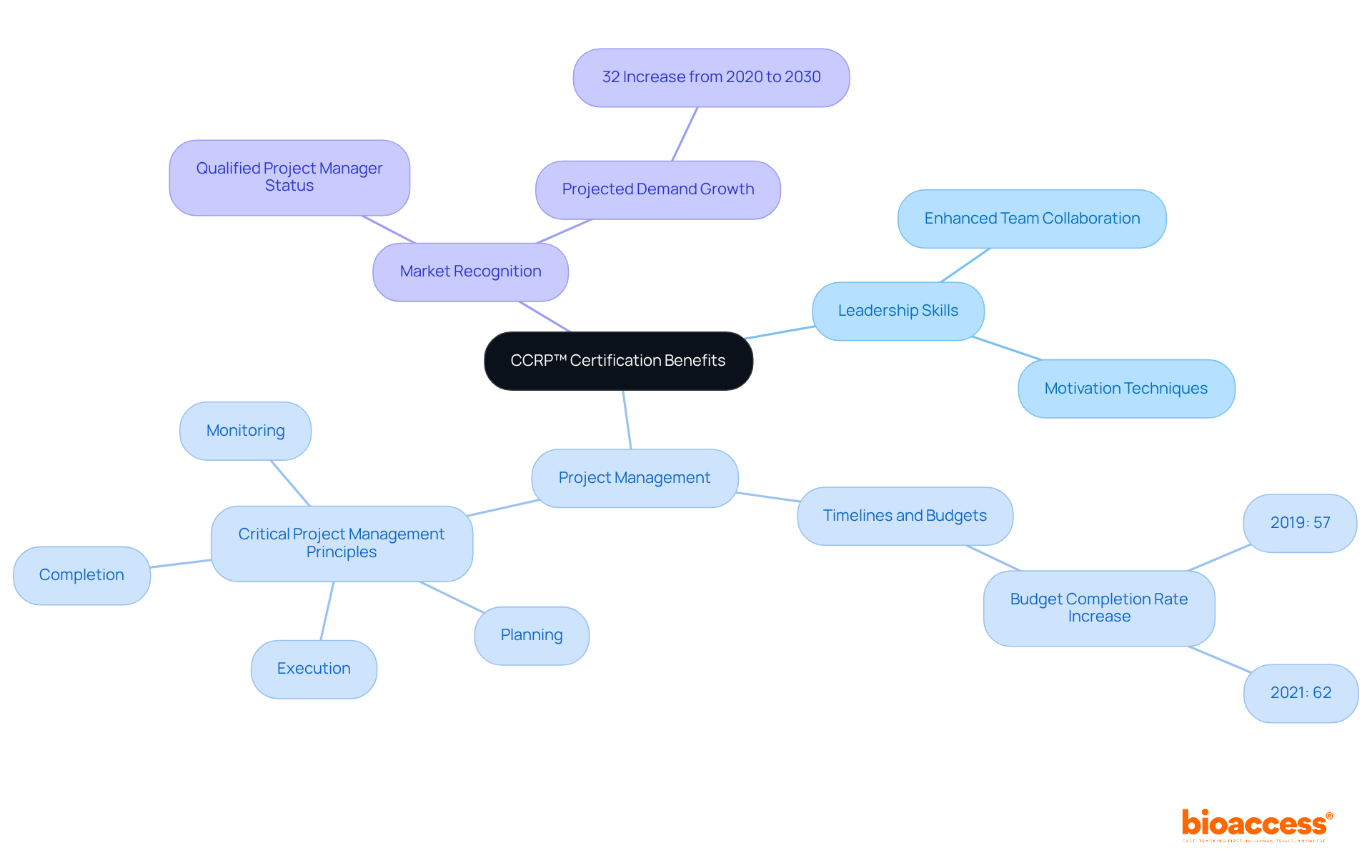
The Certified Clinical Research Project Manager (CCRP™) program equips research professionals with essential skills for effective study oversight. This certification encompasses critical project management principles, including planning, execution, monitoring, and completion of healthcare studies, ensuring a comprehensive understanding of the process.
Key benefits of the CCRP™ certification include:
- Enhanced leadership skills that foster team collaboration and motivation.
- Improved ability to manage timelines and budgets, which are crucial for project success; studies indicate that the percentage of projects completed within budget increased from 57% to 62% between 2019 and 2021.
- Recognition as a qualified project manager in the medical research sector, a distinction becoming increasingly significant as the demand for skilled professionals is projected to rise by 32% from 2020 to 2030.
Directors who obtain this certification will be better positioned to guide their teams effectively, ensuring successful study outcomes and advancing medical knowledge through well-managed research.
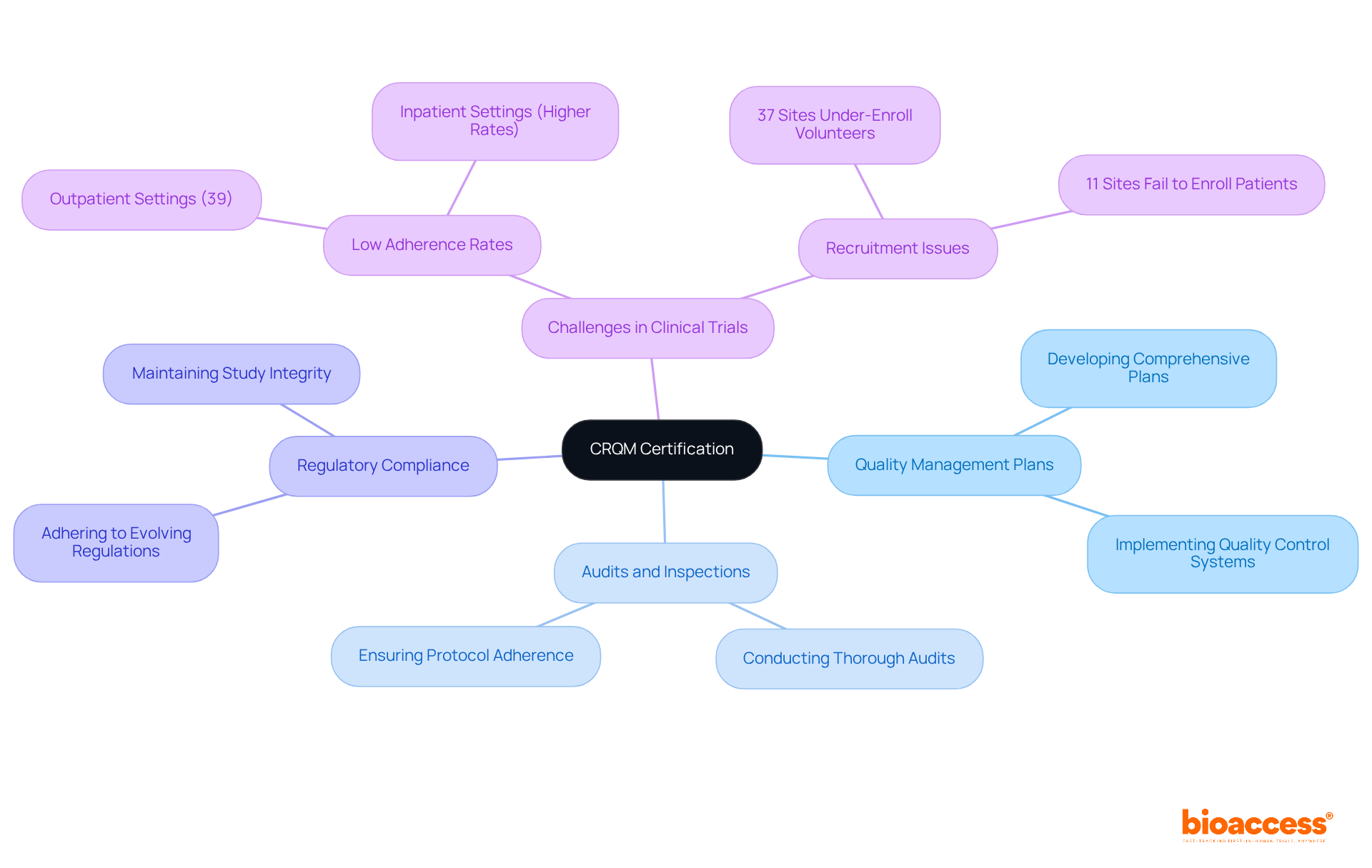
Clinical Research Quality Manager (CRQM) Certification: Validate Your Commitment to Quality
The Clinical Research Quality Manager (CRQM) certification stands as a cornerstone for professionals dedicated to upholding the highest quality standards in research studies. This certification underscores the implementation of robust quality control systems, strict adherence to regulatory compliance, and the establishment of continuous improvement processes.
Key components of effective quality management in clinical trials encompass:
- Developing and implementing comprehensive quality management plans.
- Conducting thorough audits and inspections to ensure adherence to protocols.
- Ensuring compliance with evolving regulatory requirements, which are critical for preserving the integrity of the study.
Current adherence levels in research studies reveal that, despite numerous experiments striving for excellence, significant obstacles remain. For example, adherence rates can fluctuate dramatically, with some studies reporting as low as 39% in outpatient settings, contrasted with higher rates observed in inpatient environments. By completing the clinical research management course and obtaining the CRQM certification, directors can effectively guide their teams in fostering a culture of quality and compliance, ultimately enhancing study outcomes and contributing to the advancement of medical knowledge.
Antidote Clinical Trial Project Management Courses: Enhance Your Management Skills
Antidote offers a comprehensive selection of courses, including a clinical research management course, aimed at enhancing the competencies of medical research professionals. These courses delve into essential subjects such as budgeting, resource allocation, and effective stakeholder communication, all of which are pivotal for the successful execution of health studies.
Key features of these courses encompass:
- Interactive learning modules that promote engagement and practical application
- Real-world case studies that illustrate effective project oversight strategies in healthcare studies
- Networking opportunities with industry professionals that facilitate valuable connections and knowledge exchange
By enrolling in a clinical research management course, leaders can refine their management skills, leading to improved outcomes and heightened efficiency in research operations. Current enrollment trends reflect an increasing acknowledgment of the significance of these competencies, underscoring the necessity of such training in today’s competitive environment.
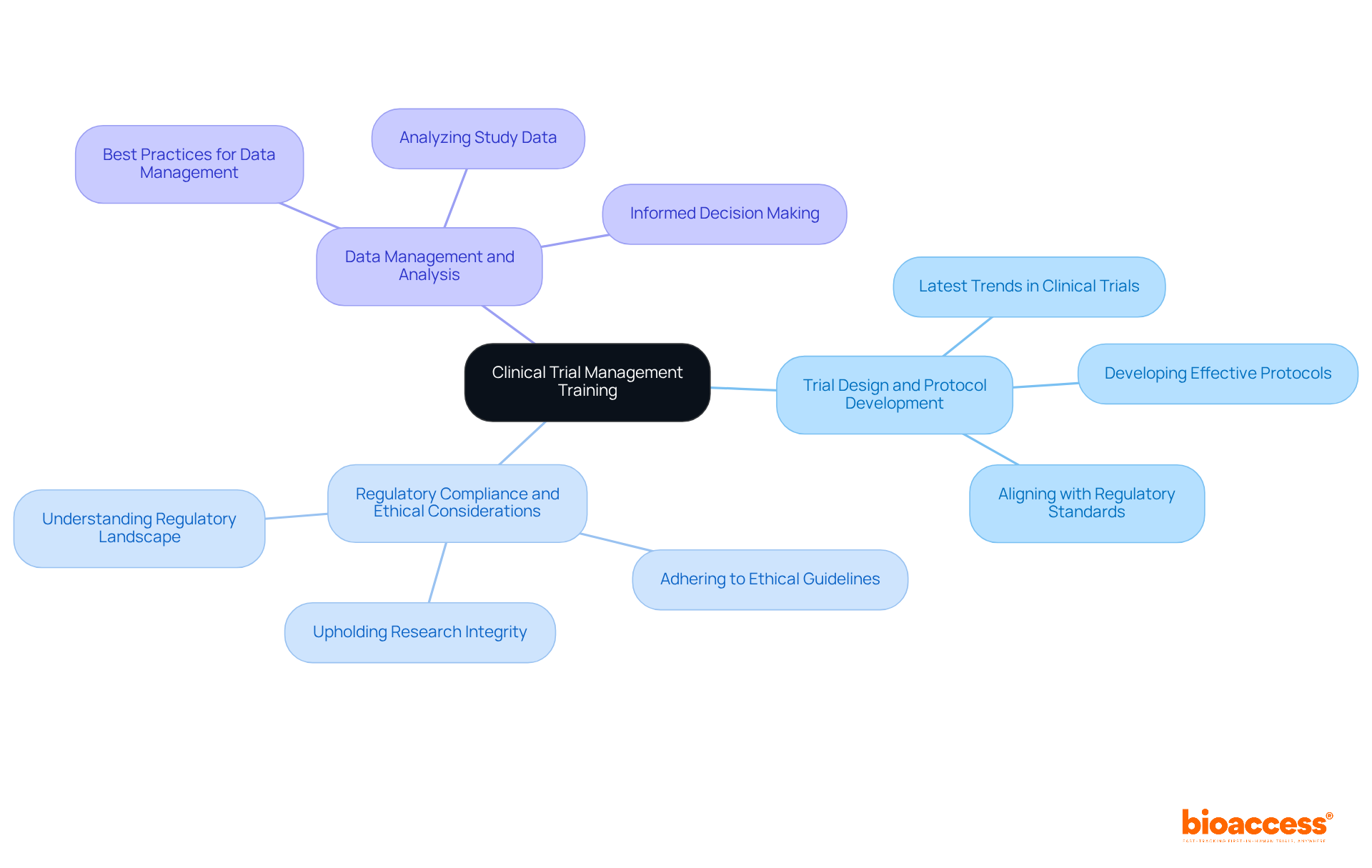
Clinical Trial Management Training: Comprehensive Skills for Success
The clinical research management course offers a comprehensive overview of the study process, equipping directors with essential skills to manage trials effectively. This training covers critical areas, including:
- Trial Design and Protocol Development: Participants will explore the latest trends in designing robust clinical trials and developing effective protocols that align with regulatory standards.
- Regulatory Compliance and Ethical Considerations: A solid understanding of the regulatory landscape is crucial. The training emphasizes the importance of adhering to ethical guidelines and regulatory requirements to uphold the integrity of research studies.
- Data Management and Analysis: Directors will acquire insights into best practices for managing and analyzing study data, which is vital for making informed decisions throughout the research process.
Upon completing the clinical research management course, leaders will be well-prepared to oversee research studies from inception to completion, ensuring compliance and maintaining high-quality standards throughout the process. This initiative aligns with ongoing efforts to enhance the skills of clinical study leaders, particularly those in the early phases of multicenter research, thereby improving the quality and efficiency of complex clinical investigations.
Early-Phase Clinical Research Training: Specialize in First-in-Human Studies
Early-Phase Clinical Research Training addresses the unique challenges and factors involved in first-in-human studies, making it essential for managers overseeing these experiments. This training is pivotal in navigating the complexities of clinical research, particularly in the Medtech landscape.
Key components of this training include:
- Safety Assessments and Ethical Considerations: Understanding the complexities of safety evaluations is crucial, particularly given the heightened scrutiny surrounding high-risk investigational products. Recent regulatory changes underscore the necessity for comprehensive safety assessments to mitigate risks associated with new therapies.
- Patient Recruitment Strategies: Effective recruitment is vital for the success of early-phase studies. Directors must implement innovative strategies to engage potential participants, ensuring timely and informed consent, which can significantly enhance recruitment rates.
- Regulatory Requirements: Familiarity with the specific regulatory landscape governing early-phase research is imperative. This involves navigating the evolving guidelines that dictate the conduct of first-in-human studies, ensuring compliance while preserving the integrity of the research.
By concentrating on early-stage medical research, leaders can broaden their knowledge, leading to improved study outcomes and enhanced patient safety.
Regulatory Compliance in Clinical Research: Essential Training for Professionals
Regulatory Compliance in Research training equips directors with the essential knowledge required to ensure that studies align with all regulatory standards. This comprehensive training covers several critical areas:
- An overview of regulatory agencies and their pivotal roles in overseeing clinical trials, including insights into the Colombian National Food and Drug Surveillance Institute (INVIMA) and its classification as a Level 4 health authority by PAHO/WHO.
- Key regulations governing research studies involving human subjects, such as Good Clinical Practice (GCP) guidelines and the Research Regulation, which underscores the necessity for ethical approval and participant safety.
- A review and feedback on study documents to ensure compliance with country requirements.
- Strategies for maintaining compliance throughout the testing process, emphasizing the establishment of robust protocols, effective data management, and ongoing staff training to adeptly navigate evolving regulations.
- The management of serious and non-serious adverse events to guarantee participant safety and adherence to regulatory standards.
By completing the clinical research management course, leaders will enhance their management skills, ensuring their teams consistently meet regulatory standards and uphold the integrity of research studies.
Managing Multiple Clinical Research Projects: Strategies for Success
Managing multiple clinical research projects necessitates a strategic approach, which is a focus of the clinical research management course, to ensure successful oversight of several trials simultaneously. This clinical research management course is essential for leaders who must navigate competing priorities and resource limitations. Key areas of focus include:
- Prioritization Techniques: Implementing effective prioritization methods enables directors to allocate resources efficiently and address the most critical tasks first. Techniques such as the Eisenhower Matrix assist in distinguishing between urgent and important tasks, ensuring that high-impact projects receive the attention they deserve.
- Effective Communication Strategies: Clear communication across teams is vital for maintaining alignment and fostering collaboration. Techniques such as regular check-ins, status updates, and the use of collaborative platforms enhance transparency and keep all stakeholders informed about project progress and challenges.
- Tools and Technologies for Project Tracking: Utilizing advanced project oversight tools streamlines tracking and coordination processes. Platforms that combine features for regulatory adherence, experiment setup, and data coordination significantly enhance oversight and efficiency in clinical studies. For instance, bioaccess offers extensive services including feasibility studies, site selection, compliance reviews, experiment setup, import permits, project oversight, and reporting. Each of these services directly supports the strategies discussed: feasibility studies assist in identifying the best locations for experiments, while project management ensures that timelines and resources are effectively administered.
By adopting these strategies and utilizing the services offered by bioaccess, leaders can significantly enhance their ability to manage multiple projects effectively, which is essential for success in a clinical research management course, resulting in improved outcomes and timely progress in research.
Clinical Trial Recruitment Strategies: Overcome Challenges in Finding Qualified Researchers
Efficient research recruitment strategies are essential for directors aiming to navigate the complexities of participant involvement. Training in this clinical research management course equips leaders with the necessary tools to directly confront recruitment challenges. Key components of this training include:
- Identifying and Engaging Target Populations: Understanding the demographics and needs of potential participants is vital. Tailored outreach can significantly amplify recruitment efforts.
- Utilizing Digital Tools for Recruitment: Harnessing technology, such as social media and online platforms, can expand reach and streamline the recruitment process. Digital tools have become increasingly indispensable, especially as many principal investigators express concerns that patients may not grasp intricate study protocols.
- Establishing Collaborations with Healthcare Providers and Community Organizations: Partnering with local healthcare organizations can foster trust and improve awareness of research studies, addressing the common issue of insufficient knowledge among potential participants. A case study highlighting the lack of awareness among potential participants illustrates how scalable video content can enhance visibility and engagement in research studies.
By mastering these strategies taught in a clinical research management course, directors can not only bolster recruitment efforts but also ensure that medical studies are adequately staffed, ultimately leading to more successful outcomes. The integration of digital recruitment tools is particularly pertinent in 2025, as trends indicate an increasing reliance on technology to enhance participant engagement and retention. As one PRIMR customer remarked, "I want you to make our trial go viral," underscoring the necessity for engaging content to tackle recruitment challenges. Furthermore, addressing the issue of rapid nurse turnover impacting enrollment will offer a more comprehensive perspective on the recruitment landscape.
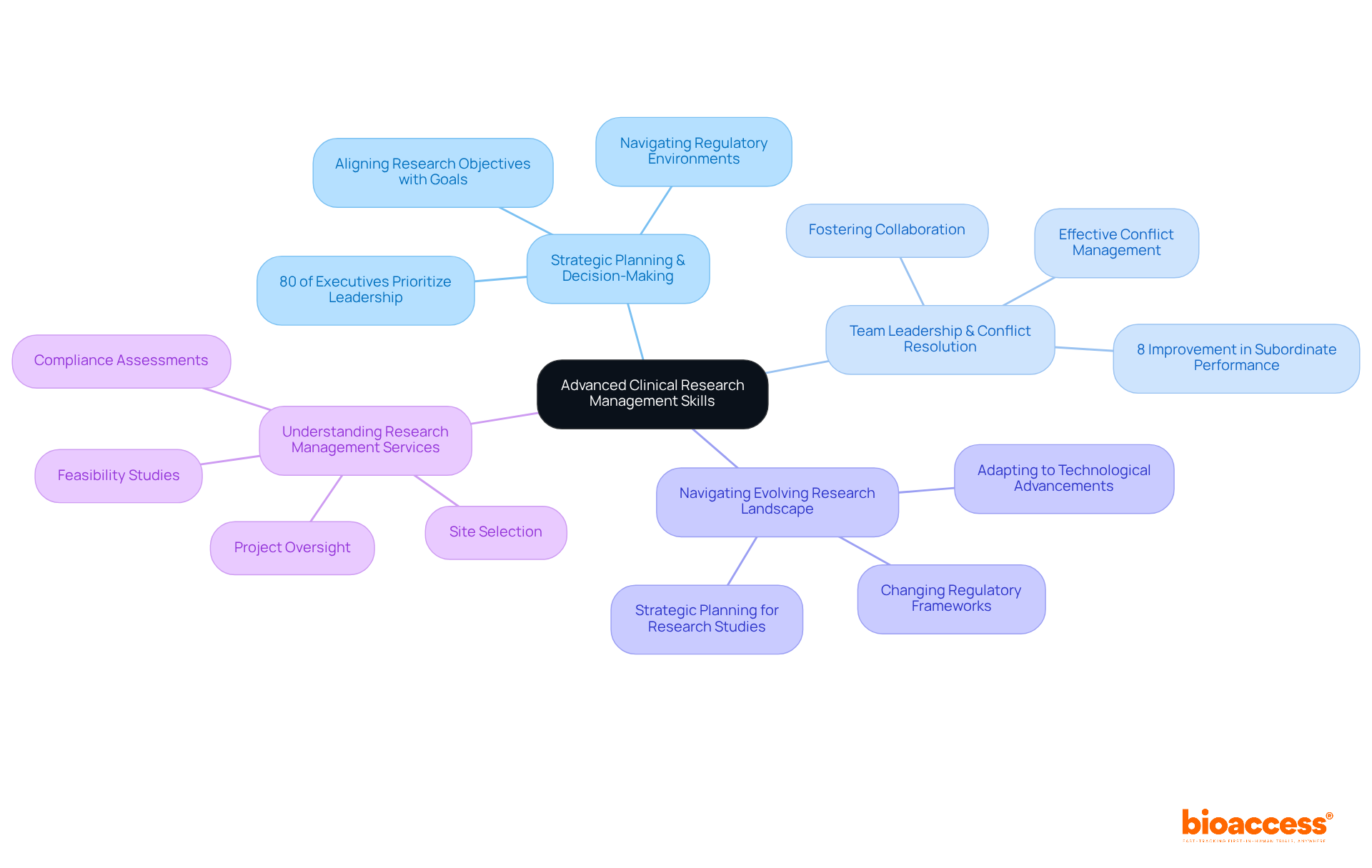
Advanced Clinical Research Management Skills: Prepare for Leadership Roles
Advanced Research Management Skills training is essential for cultivating the leadership capabilities necessary for achieving success in research. This training encompasses:
- Strategic planning and decision-making, which are vital for aligning clinical research objectives with organizational goals while navigating complex regulatory environments. Notably, 80% of executives surveyed prioritize strengthening organizational leadership, underscoring the critical nature of these skills.
- Team leadership and conflict resolution, empowering directors with the tools to foster collaboration and effectively tackle challenges within diverse teams. Research shows that subordinate performance improved by 8% following leadership training, illustrating the tangible benefits of enhancing these competencies.
- Navigating the evolving landscape of medical research, including adapting to technological advancements and changing regulatory frameworks. A pioneer in medical research once stated, "Effective strategic planning is essential for directing teams through the intricacies of research studies and ensuring successful outcomes."
In addition to these abilities, understanding extensive research management services—such as feasibility studies, site selection, compliance assessments, project initiation, import permits, project oversight, and reporting—is crucial. These components not only streamline the research process but also contribute to local economies through job creation, economic growth, healthcare improvement, and international collaboration.
By enhancing these leadership skills, directors will be better equipped to guide their teams and drive successful clinical trials, ultimately improving patient outcomes and organizational performance. To enhance your leadership skills, consider enrolling in a clinical research management course that emphasizes these essential areas.
Conclusión
The landscape of clinical research management is evolving rapidly, underscoring the critical need for specialized training among directors. Engaging in essential courses, such as those offered by bioaccess® and other reputable institutions, equips leaders with the skills necessary to navigate the complexities of modern clinical trials. These programs enhance strategic oversight and instill a culture of quality and compliance, ultimately driving improved research outcomes.
Key courses highlighted throughout the article include:
- Certified Clinical Research Project Manager (CCRP™)
- Clinical Research Quality Manager (CRQM)
Each certification is designed to furnish directors with vital project management skills and a steadfast commitment to quality. Insights into effective trial management, regulatory compliance, and innovative recruitment strategies emphasize the necessity for continuous education within this field. As the demand for skilled professionals escalates, these training opportunities become essential for career advancement and the success of clinical research initiatives.
In conclusion, investing in clinical research management courses transcends mere professional development; it constitutes a strategic imperative for directors aiming to lead successful research studies. Embracing these educational opportunities not only enhances individual capabilities but also contributes to the overall advancement of medical research, ensuring that studies are conducted efficiently and ethically. As the industry continues to evolve, proactive engagement with these training programs will position leaders at the forefront of clinical research management, ready to tackle the challenges of tomorrow.
Frequently Asked Questions
What is the bioaccess® Clinical Research Management Course?
The bioaccess® Clinical Research Management Course is designed to address the challenges of conducting research studies in regions like Latin America, Australia, and the Balkans. It focuses on regulatory efficiencies and aims to achieve ethical approvals in 4-6 weeks, faster than traditional markets.
Who is the target audience for the bioaccess® Clinical Research Management Course?
The course is tailored for leaders in clinical research who want to enhance their strategic approach to research management, emphasizing agility and efficiency.
What key topics are covered in the bioaccess® Clinical Research Management Course?
Key topics include navigating regional regulatory frameworks, innovative strategies for rapid patient recruitment, and best practices for managing multi-site studies efficiently.
What is the Certified Clinical Research Project Manager (CCRP™) program?
The CCRP™ program equips research professionals with essential project management skills for effective oversight of healthcare studies, covering planning, execution, monitoring, and completion.
What are the benefits of obtaining the CCRP™ certification?
Benefits include enhanced leadership skills, improved ability to manage timelines and budgets, and recognition as a qualified project manager in the medical research sector.
How has the percentage of projects completed within budget changed recently?
Studies indicate that the percentage of projects completed within budget increased from 57% to 62% between 2019 and 2021.
What is the Clinical Research Quality Manager (CRQM) certification?
The CRQM certification is aimed at professionals dedicated to maintaining high-quality standards in research studies, focusing on quality control systems, regulatory compliance, and continuous improvement processes.
What are the key components of effective quality management in clinical trials?
Key components include developing comprehensive quality management plans, conducting audits and inspections, and ensuring compliance with regulatory requirements.
What challenges do research studies face regarding adherence rates?
Adherence rates can vary significantly, with some outpatient studies reporting as low as 39%, while inpatient environments tend to have higher adherence rates.
How can the CRQM certification benefit directors in clinical research?
By obtaining the CRQM certification, directors can guide their teams in fostering a culture of quality and compliance, enhancing study outcomes and contributing to medical knowledge advancement.
List of Sources
- bioaccess® Clinical Research Management Course: Accelerate Your Career with Global-First Agility
- Clinical Research Regulation For Australia | ClinRegs (https://clinregs.niaid.nih.gov/country/australia)
- Number of clinical trials by year, country, region and income group (https://who.int/observatories/global-observatory-on-health-research-and-development/monitoring/number-of-clinical-trials-by-year-country-who-region-and-income-group)
- Artificial intelligence in healthcare: transforming the practice of medicine - PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC8285156)
- Rethinking Clinical Trial Country Prioritization (https://iqvia.com/insights/the-iqvia-institute/reports-and-publications/reports/rethinking-clinical-trial-country-prioritization)
- None (https://coapcr.org)
- Certified Clinical Research Project Manager (CCRP™): Master Project Management in Clinical Trials
- Understanding the Role of a Clinical Project Manager (https://invensislearning.com/blog/clinical-project-manager)
- 10 Project Management Stats All PMPs Should Know for 2022 | PMTraining (https://pmtraining.com/about/10-project-management-stats-all-pmps-should-know-for-2022)
- 70 Research Quotes to Inspire Your Work - Qualtrics (https://qualtrics.com/blog/research-quotes)
- The Pathway to Becoming a Clinical Research Associate (https://about.citiprogram.org/blog/the-pathway-to-becoming-a-clinical-research-associate)
- 139 Project Management Quotes to Inspire Your Next Project (https://plaky.com/blog/project-management-quotes)
- Clinical Research Quality Manager (CRQM) Certification: Validate Your Commitment to Quality
- What clinical trial statistics tell us about the state of research today (https://antidote.me/blog/what-clinical-trial-statistics-tell-us-about-the-state-of-research-today)
- Clinical Trials Statistics and Facts (2025) (https://media.market.us/clinical-trials-statistics)
- Protocol adherence rates in superiority and noninferiority randomized clinical trials published in high impact medical journals - PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC7529954)
- Current practice in the measurement and interpretation of intervention adherence in randomised controlled trials: A systematic review (https://sciencedirect.com/science/article/pii/S1551714422001148)
- Antidote Clinical Trial Project Management Courses: Enhance Your Management Skills
- The Community Foundation Northern Ireland - local charitable foundation (https://communityfoundationni.org/?cat=case_studies)
- Course Descriptions | Master of Public Health | School of Public Health | SUNY Downstate (https://downstate.edu/education-training/school-of-public-health/programs/master-of-public-health/course-descriptions.html)
- ITMAT Education Courses | ITMAT Education and Training | Perelman School of Medicine at the University of Pennsylvania (https://itmat.upenn.edu/itmat-ed/itmat-education-courses.html)
- Clinical Research Certification & Training Courses | CCRPS (https://ccrps.org)
- Clinical Trial Management Training: Comprehensive Skills for Success
- Call for Applications: Training Program for Multicenter Clinical Trial Management – Institute for Clinical and Translational Research (https://ictr.johnshopkins.edu/news_announce/call-for-applications-training-program-for-multicenter-clinical-trial-management)
- Applications for TIN Training Program for Multicenter Clinical Trial Management: Due MAY 30, 2025 | Institute for Translational Medicine and Science | Rutgers University (https://ritms.rutgers.edu/news/applications-for-tin-training-program-for-multicenter-clinical-trial-management-due-may-30-2025)
- Early-Phase Clinical Research Training: Specialize in First-in-Human Studies
- Phases of Clinical Trials (https://cancer.org/cancer/managing-cancer/making-treatment-decisions/clinical-trials/phases-of-clinical-trials.html)
- The RECOVERY Trial's IT team (https://recoverytrial.net/case_studies/the-recovery-trial-it-team)
- Preparing for first-in-man studies: the challenges for translational immunology post-TGN1412 - PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC2276942)
- Learning on the job: a clinician’s first experience of clinical trial research (https://recoverytrial.net/case_studies/learning-on-the-job-a-clinician2019s-first-experience-of-clinical-trial-research)
- Strategies to identify and mitigate risks for first-in-human and early clinical trials with investigational medicinal products - Scientific guideline | European Medicines Agency (EMA) (https://ema.europa.eu/en/strategies-identify-mitigate-risks-first-human-early-clinical-trials-investigational-medicinal-products-scientific-guideline)
- Regulatory Compliance in Clinical Research: Essential Training for Professionals
- 7 Key Stats on Clinical Data Management in Pharma Today (https://numberanalytics.com/blog/clinical-data-management-pharma-7-stats)
- The Role of Regulatory Affairs in Clinical Research | ICON news and blogs (https://careers.iconplc.com/blogs/2024-1/the-role-of-regulatory-affairs-in-clinical-research)
- The Importance of Regulatory Compliance in Clinical Trials (https://lindushealth.com/blog/the-importance-of-regulatory-compliance-in-clinical-trials)
- Clinical Trials Regulation | European Medicines Agency (EMA) (https://ema.europa.eu/en/human-regulatory-overview/research-development/clinical-trials-human-medicines/clinical-trials-regulation)
- Managing Multiple Clinical Research Projects: Strategies for Success
- How We Achieved a 95% Customer Retention Rate as a CRO? Expert Insights and Best Practices - NoyMed CRO (https://noymed.com/how-we-achieved-a-95-customer-retention-rate-as-a-cro-expert-insights-and-best-practices)
- EPI/PERT White Paper — Canopy Cancer Collective (https://canopycancer.org/epi-pertwhitepaper)
- DOCR Policies and Procedures | Duke University School of Medicine (https://medschool.duke.edu/research/research-support/research-support-offices/duke-office-clinical-research-docr/docr-policies)
- WCG Clinical Services: Improving Clinical Trial Quality & Efficiency (https://wcgclinical.com)
- 7 Software Clinical Trials Solutions to Streamline Research (https://bioaccessla.com/blog/7-software-clinical-trials-solutions-to-streamline-research)
- Clinical Trial Recruitment Strategies: Overcome Challenges in Finding Qualified Researchers
- Why is Clinical Trial Recruitment so Hard? - PRIMR (https://primrmed.com/blog-post/why-is-clinical-trial-recruitment-so-hard)
- Advanced Clinical Research Management Skills: Prepare for Leadership Roles
- Cancer Research UK Impact Report: management development for new and future managers - The Management Centre (https://managementcentre.co.uk/case_studies/cancer-research-uk-impact-report-management-development-for-new-and-future-managers)
- 24 Leadership Training Statistics for 2025: Data, Insights & Predictions | Research.com (https://research.com/careers/leadership-training-statistics)
- Leadership Effectiveness in Healthcare Settings: A Systematic Review and Meta-Analysis of Cross-Sectional and Before–After Studies - PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC9518077)











