


In the rapidly evolving landscape of clinical research, Romania has emerged as a pivotal player. With a robust legal framework and a diverse patient demographic, it enhances its appeal for clinical trial sponsors. This article serves as a comprehensive guide to mastering the clinical trial registry submission process in Romania. It offers insights into the regulatory requirements and best practices essential for success. However, with the impending shift to the Clinical Trials Information System (CTIS) by January 2025, stakeholders must ask: how can they effectively navigate this new terrain while ensuring compliance and efficiency in their submissions?
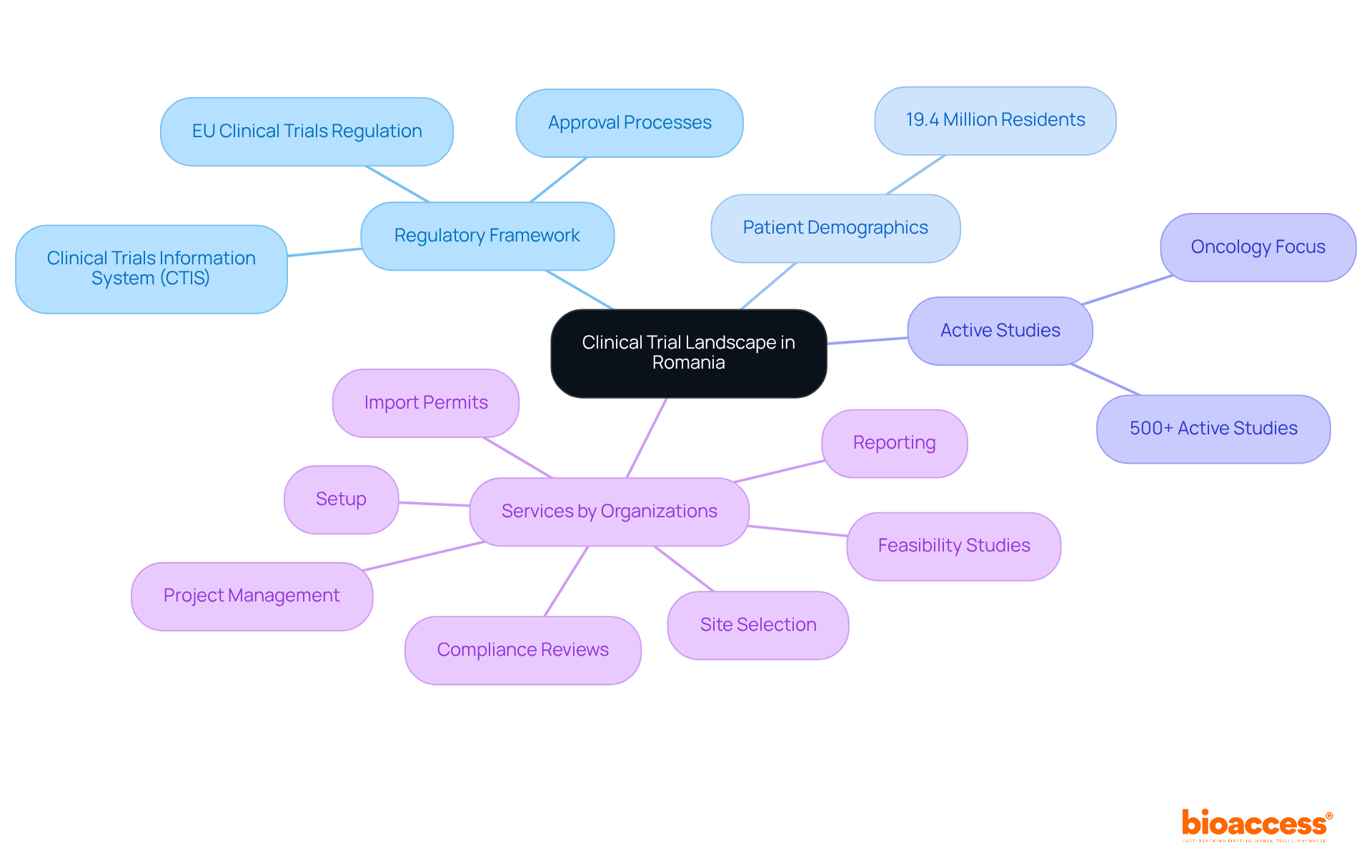
This country stands out as a key player in the clinical trial environment, particularly due to its clinical trial registry submission in Romania, which is supported by a robust legal framework and a diverse patient demographic of 19.4 million residents. Under the EU Clinical Trials Regulation (EU No. 536/2014), the country has optimized its approval processes, significantly enhancing transparency and efficiency. Beginning January 2025, all proposals must be processed through the Clinical Trials Information System (CTIS), which mandates a 60-day response period for applications. This regulatory clarity, along with Romania's competitive operational expenses and access to a large patient base, makes it an appealing location for sponsors looking to conduct clinical trial registry submission in Romania and carry out studies efficiently.
With over 500 active studies, oncology represents a major focus within these investigations. The synergy of a well-trained healthcare workforce and a supportive infrastructure further positions the country as an attractive location for international medical studies, especially for clinical trial registry submission in Romania. This environment facilitates quicker recruitment timelines and high-quality data collection. Organizations like bioaccess enhance this landscape by providing extensive management services for studies, including:
Essential elements for navigating the complexities of studies in this country.
In the evolving Medtech landscape, collaboration is crucial. By leveraging the advantages of the country and the expertise of organizations like bioaccess, stakeholders can effectively tackle significant challenges in clinical studies. The next steps involve engaging with local partners to maximize the potential of this promising research environment.
Understanding the compliance landscape is crucial before filing a clinical trial registry submission in Romania. This knowledge not only ensures adherence to regulations but also streamlines the clinical trial registry submission in Romania. Key requirements include:
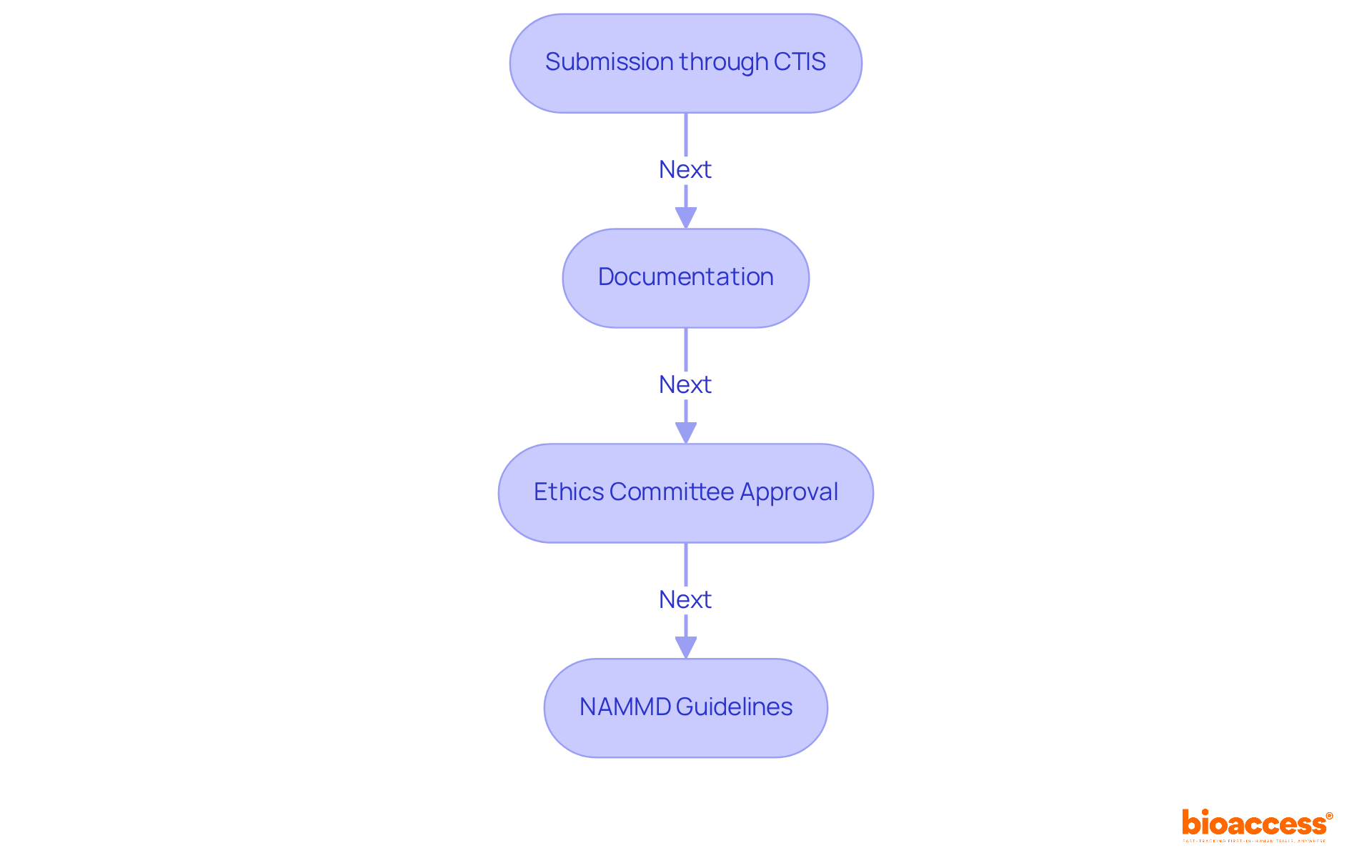
Submission through CTIS: All applications must be submitted via the Clinical Trials Information System (CTIS). This process requires prior registration and strict adherence to established guidelines to ensure compliance.
Documentation: Essential documents for submission include the clinical trial protocol, informed consent forms, and qualifications of the investigators. It is vital that all documents are either in Romanian or accompanied by certified translations to meet regulatory standards.
Ethics Committee Approval: Securing consent from a recognized ethics committee is essential before submission. This step guarantees that the procedure meets ethical standards and safeguards participant rights.
National Agency for Medicines and Medical Devices (NAMMD): Familiarity with NAMMD's guidelines is critical, as this agency oversees the approval process and ensures compliance with EU regulations. The NAMMD validates submissions and provides feedback within a structured timeline, enhancing the predictability of the approval process.
To successfully submit a clinical trial registry application in Romania, it is essential to prepare the following documentation:
Clinical Study Protocol: This document must provide a detailed description of the research's objectives, design, methodology, statistical considerations, and ethical considerations. It serves as the foundation for the assessment, guiding its execution and ensuring compliance with regulatory standards.
Informed Consent Forms: These documents are crucial as they outline the research's purpose, procedures, risks, and benefits, ensuring that participants fully understand what their involvement entails. Clear and comprehensive informed consent forms are vital for ethical compliance and participant engagement. Experts emphasize that the readability and clarity of these forms significantly impact participant comprehension and willingness to participate. Studies indicate that 76.3% of informed consent forms analyzed had poor readability, which can hinder participant understanding. Effective communication of risks and benefits is crucial for ethical conduct, especially concerning clinical trial registry submission in Romania, and forms should ideally not exceed six pages to maintain clarity and engagement.
Investigator's Brochure: This compilation includes both clinical and non-clinical data on the investigational product, providing essential information to investigators about the product's safety and efficacy.
Ethics Committee Approval Letter: This letter serves as documentation validating that the project has been reviewed and sanctioned by an ethics committee, ensuring that ethical standards are upheld throughout the inquiry process.
Financial Disclosure: This section must include information regarding any financial interests or arrangements that could influence the conduct of the proceedings, promoting transparency and integrity in the research.
Site Information: Comprehensive details about the research locations are needed, including the qualifications of the researchers and the facilities available for conducting the research. This guarantees that the examination is carried out in a setting that complies with regulatory and safety standards.
Informed consent documents, specifically, must be created to enhance understanding, as research shows that poorly constructed forms can obstruct participant comprehension. Utilizing bioaccess's extensive study management services, encompassing feasibility assessments and site selection, can greatly improve the efficiency of the application process. With an emphasis on enhancing patient recruitment and guaranteeing FDA-prepared data delivery, bioaccess offers a cost-to-speed benefit that can assist in addressing typical obstacles encountered in early-phase research.

To effectively navigate the submission process on the Clinical Trials Information System (CTIS) platform in Romania, follow these essential steps:
Staying informed about the latest CTIS regulations is crucial for ensuring compliance and avoiding common application mistakes related to clinical trial registry submission in Romania. Romania's efficient governing environment, characterized by lower operational costs and a skilled medical workforce, makes it an attractive location for clinical trial registry submission in Romania. However, navigating the CTIS can present challenges, including the need for meticulous documentation and adherence to specific formatting requirements. The deadline for a response after sending is 60 days, underscoring the importance of prompt and precise entries. Collaborating with skilled compliance experts at bioaccess can simplify this process, ensuring that all applications meet the required criteria for approval.

During the filing process, you may encounter several common issues that can hinder your progress. Here are some essential troubleshooting tips to navigate these challenges effectively:
Document Rejections: If documents are rejected, carefully examine the feedback provided by the governing authority. Ensure that all necessary documents are included and properly formatted. Bioaccess offers extensive assistance in reviewing and providing feedback on research documents to meet country requirements, ensuring your submissions are compliant and complete.
Technical Issues with CTIS: Should you experience technical difficulties with the CTIS platform, first check for system updates or maintenance notifications. If issues persist, do not hesitate to contact technical support. Bioaccess can assist with trial setup and start-up processes, ensuring smooth operations and minimizing disruptions.
Delayed Replies: If you do not receive a reply within the anticipated timeframe, it’s crucial to follow up with the governing body to inquire about the status of your submission. Bioaccess's project management services can enhance communication with governing authorities, ensuring you stay informed and proactive.
Incomplete Applications: Ensure that all sections of the application are filled out completely. Missing information can lead to significant delays. Bioaccess's expertise in feasibility assessments and site selection can help guarantee that applications are thorough and complete, reducing the risk of rejection.
Communication Gaps: Maintain clear communication with all stakeholders involved in the study, including ethics committees and regulatory bodies. This ensures everyone is aligned and informed. Bioaccess emphasizes the importance of effective communication throughout the trial management process, fostering collaboration and efficiency.

Mastering the clinical trial registry submission process in Romania is crucial for stakeholders looking to conduct research in this dynamic environment. Understanding the regulatory landscape and leveraging available resources allows sponsors to navigate the complexities of clinical trials more effectively, ensuring compliance and enhancing the likelihood of successful outcomes.
This guide highlights key aspects such as:
Preparing comprehensive and clear informed consent forms is essential, as is adhering to ethical standards and maintaining open communication with regulatory authorities to facilitate smoother submissions. The support of organizations like bioaccess is invaluable in optimizing these processes, making Romania an attractive site for clinical research.
As the clinical trial landscape in Romania evolves, staying informed and proactive is vital for success. Engaging with local experts and understanding the intricacies of submission requirements can significantly streamline the process. For those involved in clinical research, embracing these strategies will not only enhance operational efficiency but also contribute to advancing medical knowledge and improving patient outcomes in Romania and beyond.
What makes Romania a key player in the clinical trial landscape?
Romania is notable for its clinical trial registry submission, supported by a robust legal framework and a diverse patient demographic of 19.4 million residents. The country has optimized its approval processes under the EU Clinical Trials Regulation, enhancing transparency and efficiency.
What is the Clinical Trials Information System (CTIS)?
The Clinical Trials Information System (CTIS) is a platform that mandates all clinical trial proposals in Romania to be processed through it starting January 2025. It requires a 60-day response period for applications.
How many active studies are there in Romania, and what is a major focus area?
There are over 500 active studies in Romania, with oncology being a major focus area within these investigations.
What advantages does Romania offer for conducting clinical trials?
Romania offers competitive operational expenses, access to a large patient base, a well-trained healthcare workforce, and a supportive infrastructure, which facilitate quicker recruitment timelines and high-quality data collection.
What services does bioaccess provide for clinical studies in Romania?
Bioaccess provides extensive management services for studies, including feasibility studies, site selection, compliance reviews, setup, import permits, project management, and reporting.
What are the key regulatory requirements for clinical trial registry submission in Romania?
Key requirements include submission through CTIS, providing essential documentation (clinical trial protocol, informed consent forms, and investigator qualifications), obtaining ethics committee approval, and familiarity with guidelines from the National Agency for Medicines and Medical Devices (NAMMD).
What documentation is necessary for a clinical trial registry submission in Romania?
Necessary documentation includes the clinical trial protocol, informed consent forms, and qualifications of the investigators. All documents must be in Romanian or accompanied by certified translations.
Why is ethics committee approval important in the submission process?
Ethics committee approval is essential to ensure that the clinical trial meets ethical standards and safeguards the rights of participants before submission.
What role does the National Agency for Medicines and Medical Devices (NAMMD) play in the approval process?
NAMMD oversees the approval process, ensures compliance with EU regulations, validates submissions, and provides feedback within a structured timeline, enhancing the predictability of the approval process.